2
answers
0
watching
31
views
18 Nov 2019
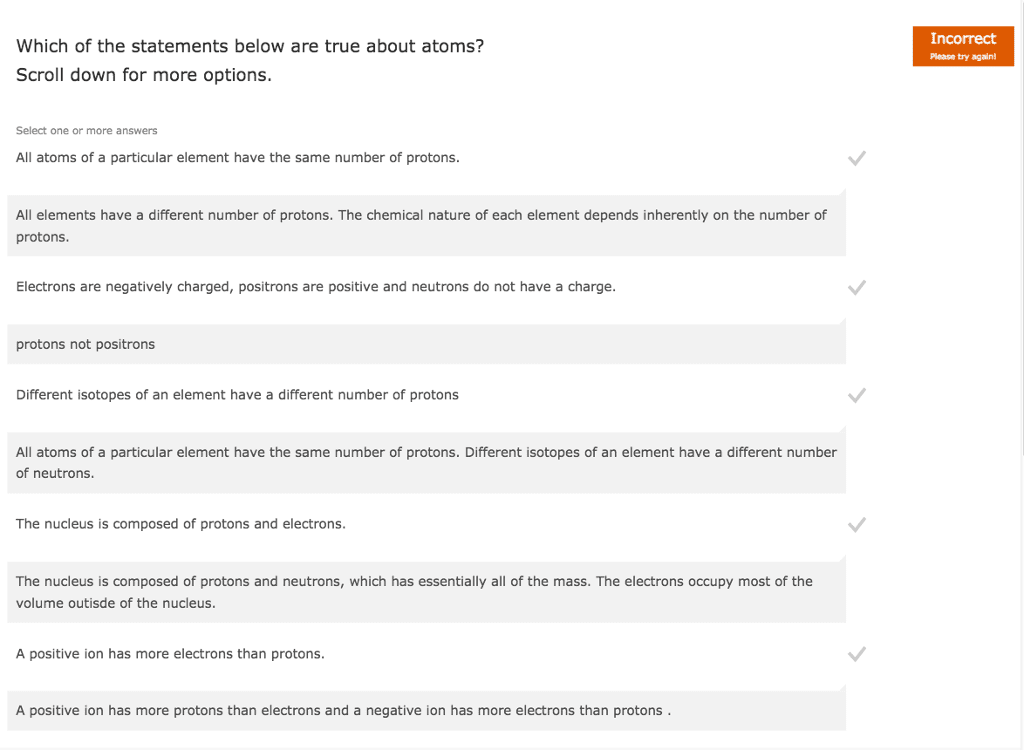
True or false?
A. All isotopes of an element have the same number of protons
B. There are more nonmetals than metals in the periodic table.
C. A scientific theory can never change.
D. Except for hydrogen, all elements have neutrons in their nuclei.
E. The element with 30 protons in the nucleus is Zn.
F. The noble gas Ar has the same number of electrons as a S2- ion.
G. In a diatomic molecule the bonding electrons are closer to the atom with the higher electronegativity.
H. Ionic compounds have a net electrical charge.
I. When involved in ionic bonding the group 7A elements gain one electron per atom.
J. Compounds composed of nonmetals have covalent bonding.
True or false?
A. All isotopes of an element have the same number of protons
B. There are more nonmetals than metals in the periodic table.
C. A scientific theory can never change.
D. Except for hydrogen, all elements have neutrons in their nuclei.
E. The element with 30 protons in the nucleus is Zn.
F. The noble gas Ar has the same number of electrons as a S2- ion.
G. In a diatomic molecule the bonding electrons are closer to the atom with the higher electronegativity.
H. Ionic compounds have a net electrical charge.
I. When involved in ionic bonding the group 7A elements gain one electron per atom.
J. Compounds composed of nonmetals have covalent bonding.
simipster332Lv1
16 Dec 2022
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Tod ThielLv2
19 Jan 2019
Get unlimited access
Already have an account? Log in