1
answer
2
watching
573
views
manya12Lv1
11 Jun 2021
Show all your work
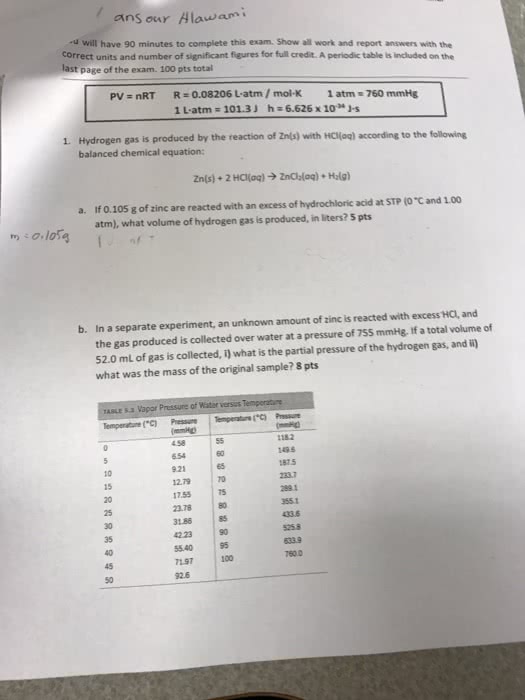
Write the reaction between solid zinc (II) bisulfite and aqueous hydroiodic acid.
Assume ∆H = -500.0 KJ/mol zn (II) bisulfite, and the reaction occurs under constant pressure.
During this reaction a gas was produced. If 200. m3 of gas was produced at a temperature of 98.0⁰F and 1.07 atm, calculate the amount of heat (KJ) produced.
Show all your work
Write the reaction between solid zinc (II) bisulfite and aqueous hydroiodic acid.
Assume ∆H = -500.0 KJ/mol zn (II) bisulfite, and the reaction occurs under constant pressure.
During this reaction a gas was produced. If 200. m3 of gas was produced at a temperature of 98.0⁰F and 1.07 atm, calculate the amount of heat (KJ) produced.
Liked by manya12 and 6 others
11 Jun 2021