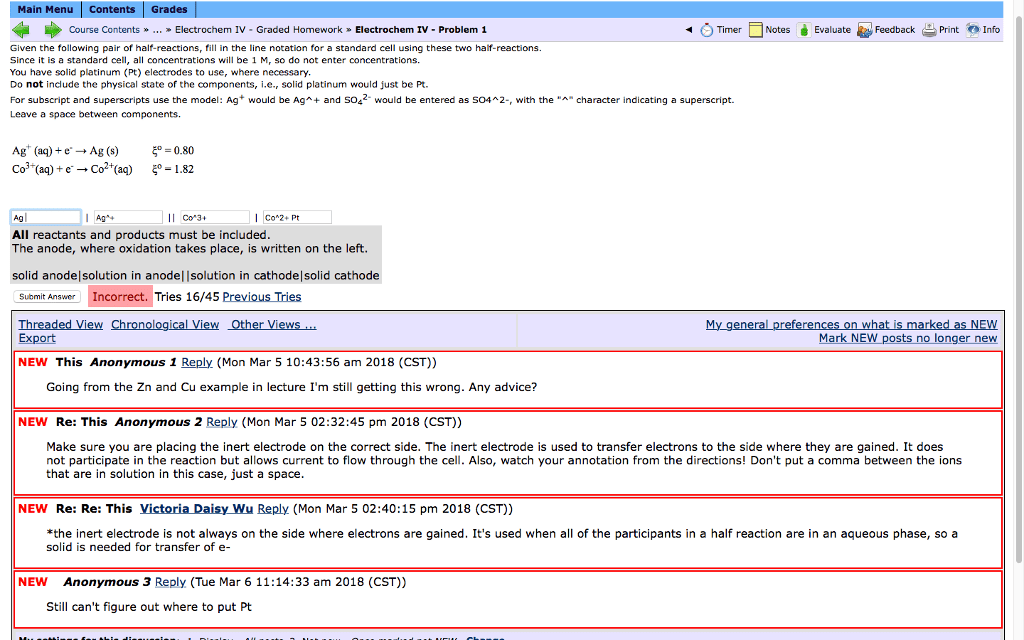
I need help solving the following Physical chemistry kinetics problem using Wolfram Mathematica please. Please see the attached documents.
I would really appreciate your help.
Problem 1. Assume that the reaction
Subscript[O, 3] + M -> 3/2 Subscript[O, 2] + M
is elementary and write down a plausible rate equation for Subscript[dO, 3](t)/dt. Subscript[O, 3](t) is the concentration of ozone (mol/liter) at time t.
Problem 2. Assume that you know that the reaction takes place in two steps
Subscript[O, 3] + M \[RightArrowLeftArrow] Subscript[O, 2] + O + M (1)
and
O + Subscript[O, 3] -> 2 Subscript[O, 2] (2)
Here M is an inert molecule, such as nitrogen or argon. Write rate equations for all compounds in reaction; express Subscript[dO, 3]/dt, dO/dt and Subscript[dO, 2]/dt, in terms of the concentrations of Subscript[O, 2], Subscript[O, 3], O, and M.
Problem 3. Write the rate equations for the reactions 1 and 2 (given in problem 2) in terms of the extents of reactions.
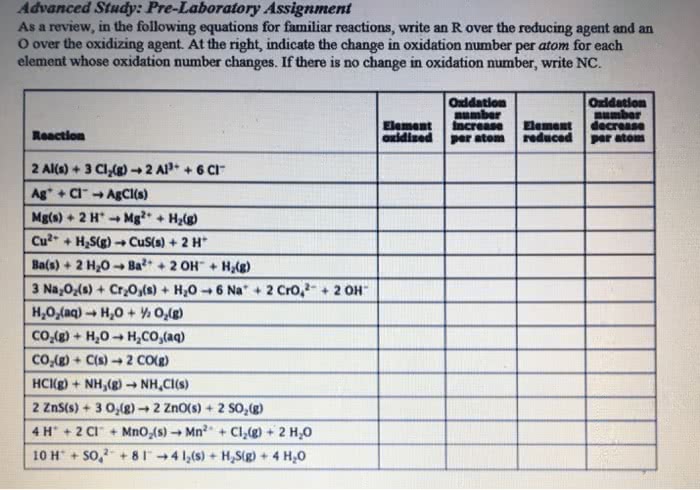
I need help solving the following Physical chemistry kinetics problem using Wolfram Mathematica please. Please see the attached documents.
I would really appreciate your help.
Problem 1. Assume that the reaction
Subscript[O, 3] + M -> 3/2 Subscript[O, 2] + M
is elementary and write down a plausible rate equation for Subscript[dO, 3](t)/dt. Subscript[O, 3](t) is the concentration of ozone (mol/liter) at time t.
Problem 2. Assume that you know that the reaction takes place in two steps
Subscript[O, 3] + M \[RightArrowLeftArrow] Subscript[O, 2] + O + M (1)
and
O + Subscript[O, 3] -> 2 Subscript[O, 2] (2)
Here M is an inert molecule, such as nitrogen or argon. Write rate equations for all compounds in reaction; express Subscript[dO, 3]/dt, dO/dt and Subscript[dO, 2]/dt, in terms of the concentrations of Subscript[O, 2], Subscript[O, 3], O, and M.
Problem 3. Write the rate equations for the reactions 1 and 2 (given in problem 2) in terms of the extents of reactions.