2
answers
1
watching
310
views
miuLv1
13 Mar 2021
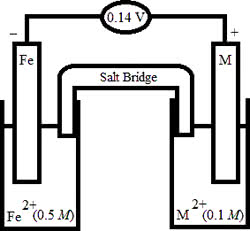
If an electrochemical cell, Rb | Rb+|| Li+| Li has a standard potential of +0.02 V, what is the standard reduction potential of the Rb half-cell if the Li+/Li half-reaction has a reduction potential of -3.05 V?
Group of anwers:
-3.03 V
+3.07 V
-3.07 V
0 V
+3.03 V
If an electrochemical cell, Rb | Rb+|| Li+| Li has a standard potential of +0.02 V, what is the standard reduction potential of the Rb half-cell if the Li+/Li half-reaction has a reduction potential of -3.05 V?
Group of anwers:
-3.03 V
+3.07 V
-3.07 V
0 V
+3.03 V
18 May 2023
2 Jun 2021
Already have an account? Log in