1
answer
0
watching
292
views
violetant532Lv1
28 Nov 2020
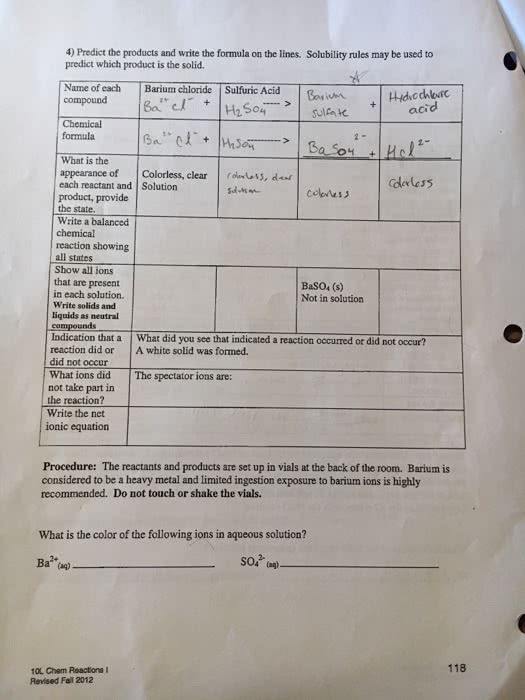
- Write an equation for the chemical reaction that occurred between sulfuric acid and barium hydroxide.
- Explain in terms of the properties of the products formed why the light went out when barium hydroxide was added to the sulfuric acid solution, even though both of these reactants are electrolytes.
- Explain why the light came on again when additional barium hydroxide was added.
- In the conductivity tests, what controlled the brightness of the light?
- Write an equation to show how acetic acid reacts with water to produce ions in solution.
- What classes of compounds tested are electrolytes?
- Write an equation for the chemical reaction that occurred between sulfuric acid and barium hydroxide.
- Explain in terms of the properties of the products formed why the light went out when barium hydroxide was added to the sulfuric acid solution, even though both of these reactants are electrolytes.
- Explain why the light came on again when additional barium hydroxide was added.
- In the conductivity tests, what controlled the brightness of the light?
- Write an equation to show how acetic acid reacts with water to produce ions in solution.
- What classes of compounds tested are electrolytes?
Robert KubaraLv10
22 Jan 2021