1
answer
0
watching
243
views
6 Oct 2020
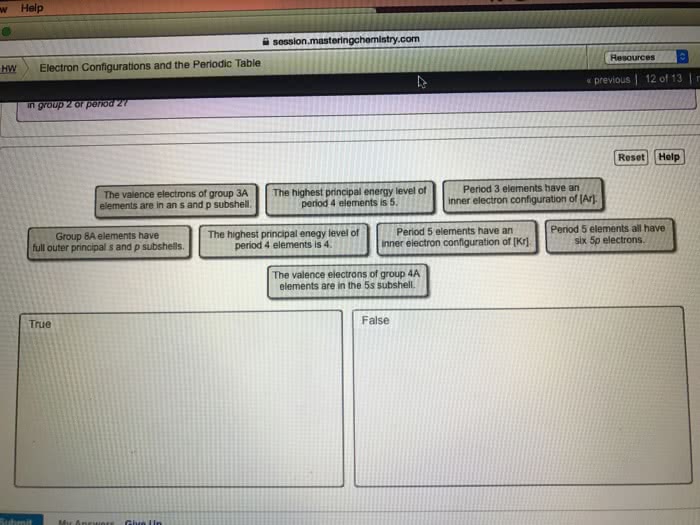
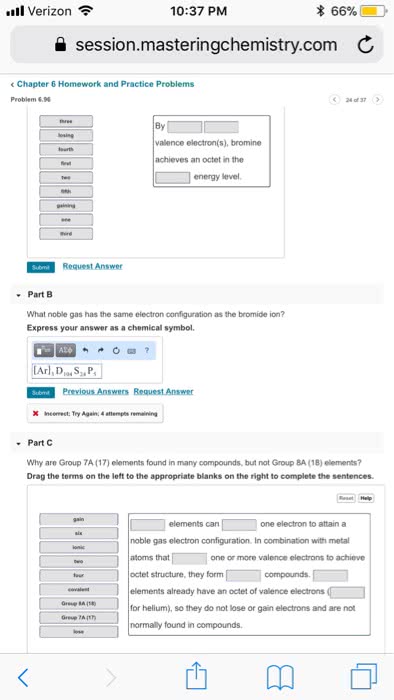
Valence electrons are those electrons in the outermost principal quantum level (highest n level) of an atom in its ground state. Groups 1A to 8A have from 1 to 8 valence electrons. For each group of the representative elements (1A-8A). Give the number of valence electrons, the general valence electron configuration, a sample element in that group, and the specific valence electron configuration for that element.
Valence electrons are those electrons in the outermost principal quantum level (highest n level) of an atom in its ground state. Groups 1A to 8A have from 1 to 8 valence electrons. For each group of the representative elements (1A-8A). Give the number of valence electrons, the general valence electron configuration, a sample element in that group, and the specific valence electron configuration for that element.
Robert KubaraLv10
10 Nov 2020