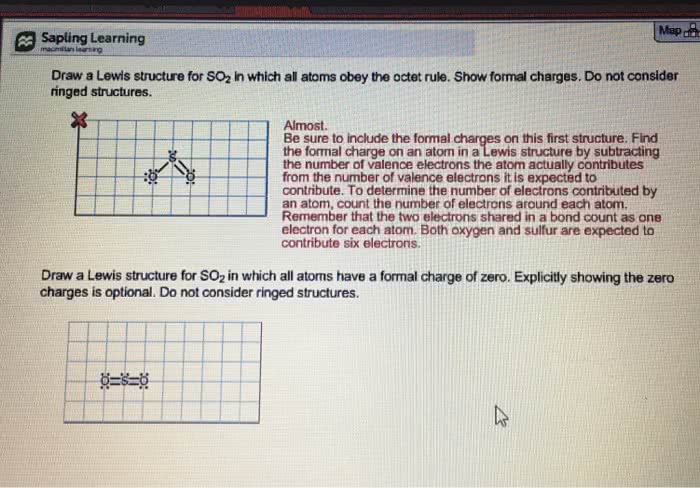
1
answer
0
watching
363
views
blushlion324Lv1
6 Oct 2020
It is possible to write a simple Lewis structure for the SO42- ion involving only single bonds, which follows the octet rule. However, Linus Pauling and others have suggested an alternative structure, involving double bonds, in which the sulfur atom is surrounded by six electron pairs.
(a) Draw the two Lewis structures?
(b) What geometries are predicted for the two Lewis structures?
(c) What is the hybridization of sulfur in each case?
(d) What are the formal charges of atoms in the two structures?
It is possible to write a simple Lewis structure for the SO42- ion involving only single bonds, which follows the octet rule. However, Linus Pauling and others have suggested an alternative structure, involving double bonds, in which the sulfur atom is surrounded by six electron pairs.
(a) Draw the two Lewis structures?
(b) What geometries are predicted for the two Lewis structures?
(c) What is the hybridization of sulfur in each case?
(d) What are the formal charges of atoms in the two structures?
Kottherva SreevidyaLv10
22 Dec 2020