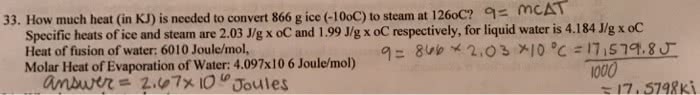1
answer
0
watching
467
views
6 Oct 2020
You have a 1.00-mole sample of water at -30 oC and you heat it until you have gaseous water at 140 oC. What is the energy absorbed by water in the whole process? Use the following data.
Specific heat capacity of ice = 2.03 J oC-1 g-1
Specific heat capacity of water = 4.18 J oC-1 g-1
Specific heat capacity of steam = 2.02 J oC-1 g-1
Heat of fusion (
Heat of vaporization (
You have a 1.00-mole sample of water at -30 oC and you heat it until you have gaseous water at 140 oC. What is the energy absorbed by water in the whole process? Use the following data.
Specific heat capacity of ice = 2.03 J oC-1 g-1
Specific heat capacity of water = 4.18 J oC-1 g-1
Specific heat capacity of steam = 2.02 J oC-1 g-1
Heat of fusion (
Heat of vaporization (
Brian HingpitLv10
17 Nov 2020