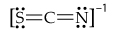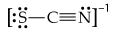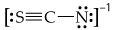1
answer
0
watching
139
views
6 Oct 2020
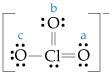
Draw the resonance structures for the bicarbonate ion, 
(a) Does 

(b) What are the formal charges on the O and C atoms in 

(c) Protonation of 


Draw the resonance structures for the bicarbonate ion, 
(a) Does 

(b) What are the formal charges on the O and C atoms in 

(c) Protonation of 


Camille Sta. MariaLv8
16 Dec 2020