1
answer
0
watching
585
views
6 Oct 2020
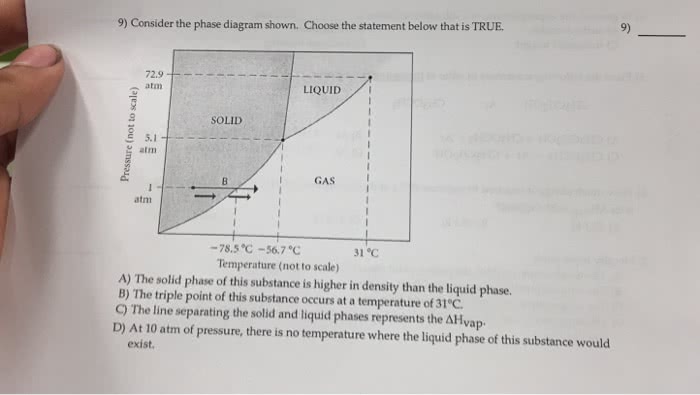
Choose the statements that correctly describe the following phase diagram.

a. If the temperature is raised from 50 K to 400 K at a pressure of 1 atm, the substance boils at approximately 185 K.
b. The liquid phase of this substance cannot exist under conditions of 2 atm at any temperature.
c. The triple point occurs at approximately 165 K.
d. At a pressure of 1.5 atm, the melting point of the substance is approximately 370 K.
e. The critical point occurs at approximately 1.7 atm and 410 K.
Choose the statements that correctly describe the following phase diagram.

a. If the temperature is raised from 50 K to 400 K at a pressure of 1 atm, the substance boils at approximately 185 K.
b. The liquid phase of this substance cannot exist under conditions of 2 atm at any temperature.
c. The triple point occurs at approximately 165 K.
d. At a pressure of 1.5 atm, the melting point of the substance is approximately 370 K.
e. The critical point occurs at approximately 1.7 atm and 410 K.
Patrick SuarezLv10
8 Dec 2020