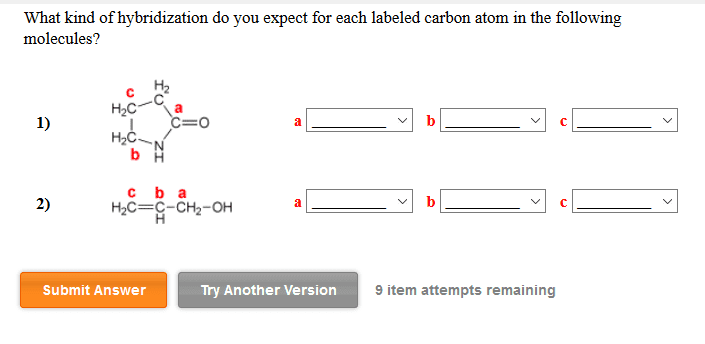
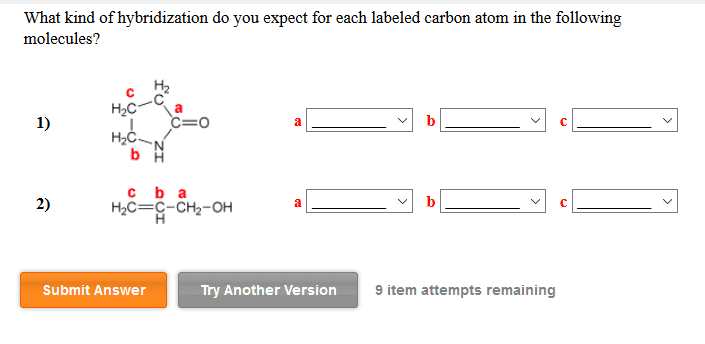
1
answer
0
watching
333
views
greymouse614Lv1
6 Oct 2020
Allene(1,2-propadiene), H2C=C=CH2 has two adjacent double bonds. What kind of hybridization must the central atom have? Sketch the bonding orbitals in allene? What shape do you predict for allene?
Allene(1,2-propadiene), H2C=C=CH2 has two adjacent double bonds. What kind of hybridization must the central atom have? Sketch the bonding orbitals in allene? What shape do you predict for allene?
Kottherva SreevidyaLv10
10 Dec 2020