1
answer
0
watching
263
views
6 Oct 2020
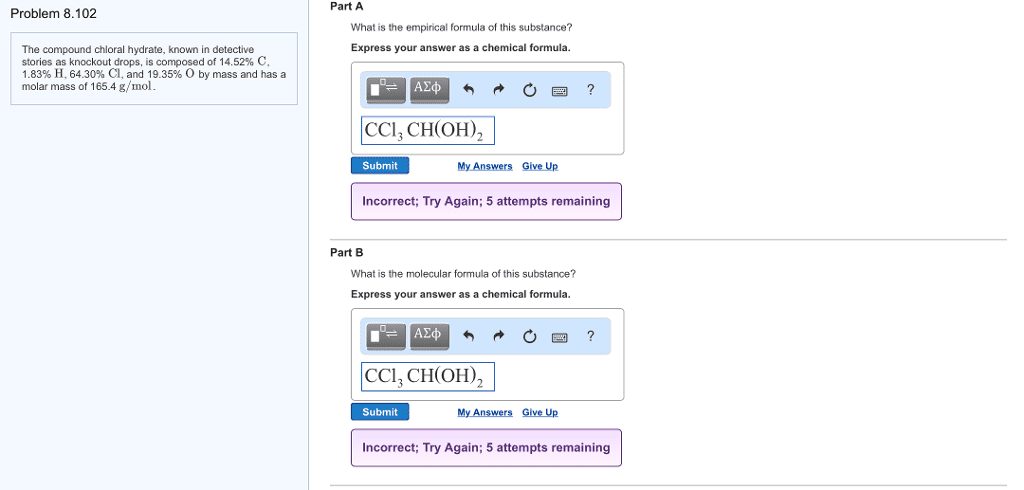
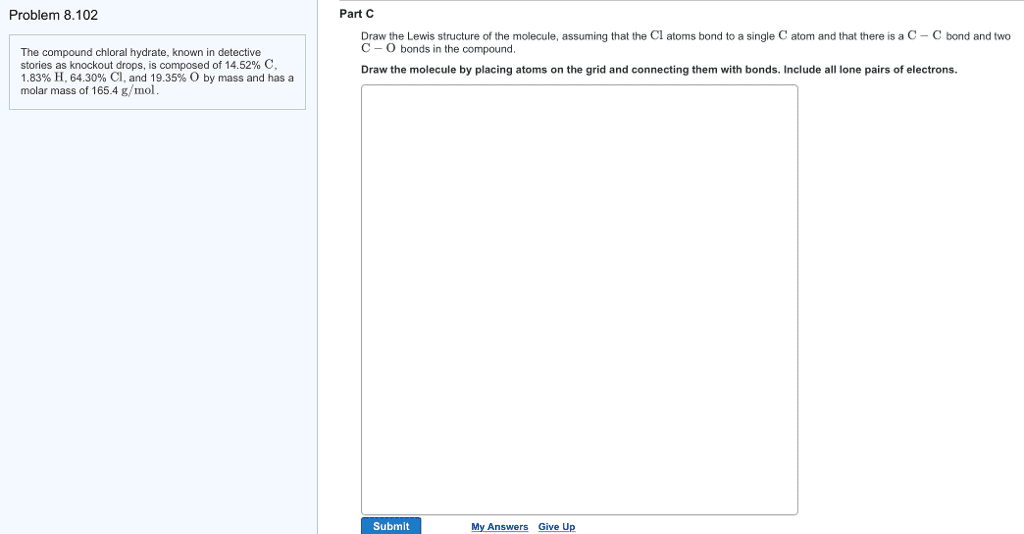
Chloral hydrate (C2H3Cl3O2) is a drug formerly used as a sedative and hypnotic. It is the compound used to make "Mickey Finns" in detective stories.
a. Calculate the molar mass of chloral hydrate.
b. What amount (moles) of C2H3Cl3O2 molecules are in 500.0 g chloral hydrate?
c. What is the mass in grams of 2.0 x 10-2 moles of chloral hydrate?
d. What number of chlorine atoms are in 5.0 g chloral hydrate?
e. What mass of chloral hydrate would contain 1.0g Cl?
f. What is the mass of exactly 500 molecules of chloral hydrate?
Chloral hydrate (C2H3Cl3O2) is a drug formerly used as a sedative and hypnotic. It is the compound used to make "Mickey Finns" in detective stories.
a. Calculate the molar mass of chloral hydrate.
b. What amount (moles) of C2H3Cl3O2 molecules are in 500.0 g chloral hydrate?
c. What is the mass in grams of 2.0 x 10-2 moles of chloral hydrate?
d. What number of chlorine atoms are in 5.0 g chloral hydrate?
e. What mass of chloral hydrate would contain 1.0g Cl?
f. What is the mass of exactly 500 molecules of chloral hydrate?
Kenneth DuqueLv10
26 Oct 2020