How does a catalyst change a potential energy diagram?
How does a catalyst change a potential energy diagram?
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
Match the terms for this potential energy diagram for an exothermic reaction to the numbers.
| Forward Activation energy | |
| Reverse Activation energy | |
| Reactants | |
| Products | |
| With Catalyst | |
| Heat of Reaction |
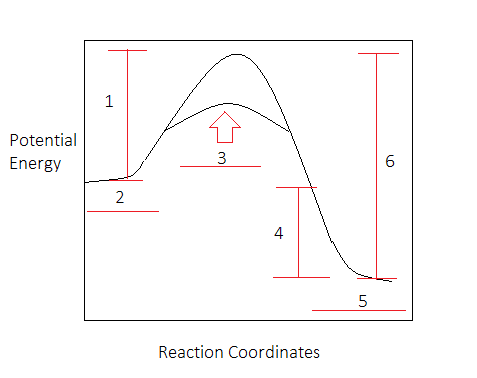


What is the value of the activation energy of the uncatalyzed reaction?
Express your answer to three significant figures and include the appropriate units.
Part B
What is the value of the enthalpy change of the uncatalyzed reaction?
Express your answer to three significant figures and include the appropriate units.
Part C
What is the value of the activation energy of the uncatalyzed reaction in reverse?
Part D
What is the value of the enthalpy change of the uncatalyzed reaction in reverse?
Express your answer to three significant figures and include the appropriate units.
Part E
How does the presence of a catalyst affect the activation energy of a reaction?
How does the presence of a catalyst affect the activation energy of a reaction?
| A. A catalyst increases the activation energy of a reaction. |
| B. A catalyst decreases the activation energy of a reaction. |
| C. A catalyst does not affect the activation energy of a reaction. |
| D. It depends on whether you are talking about the forward or the reverse reaction. |
Part F
How does the presence of a catalyst affect the enthalpy change of a reaction?
How does the presence of a catalyst affect the enthalpy change of a reaction?
| A.A catalyst increases the enthalpy change of a reaction. |
| B.A catalyst decreases the enthalpy change of a reaction. |
| C.A catalyst does not affect the enthalpy change of a reaction. |
| D.It depends on whether you are talking about the forward or the reverse reaction. |

