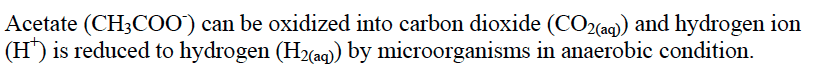1
answer
0
watching
179
views
8 Jun 2020
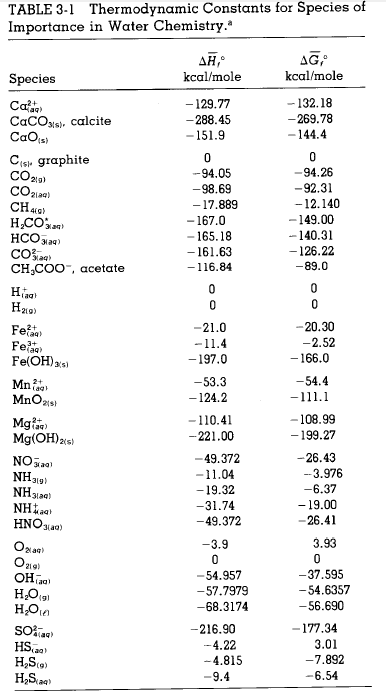
The values of  and
and  for the reaction below are
for the reaction below are  and
and  , respectively.
, respectively.

Consider the following conditions.
1. At  , the concentrations of hydrogen acetate and sodium acetate are both
, the concentrations of hydrogen acetate and sodium acetate are both  while the pressure of carbon dioxide is
while the pressure of carbon dioxide is  .
.
2. At  , the concentrations of hydrogen acetate and sodium acetate are
, the concentrations of hydrogen acetate and sodium acetate are  and
and  , respectively, while the pressure of carbon dioxide is
, respectively, while the pressure of carbon dioxide is  .
.
Given the above conditions, at which condition is sodium bicarbonate at a higher concentration?
The values of and
for the reaction below are
and
, respectively.

Consider the following conditions.
1. At , the concentrations of hydrogen acetate and sodium acetate are both
while the pressure of carbon dioxide is
.
2. At , the concentrations of hydrogen acetate and sodium acetate are
and
, respectively, while the pressure of carbon dioxide is
.
Given the above conditions, at which condition is sodium bicarbonate at a higher concentration?
1 Sep 2020