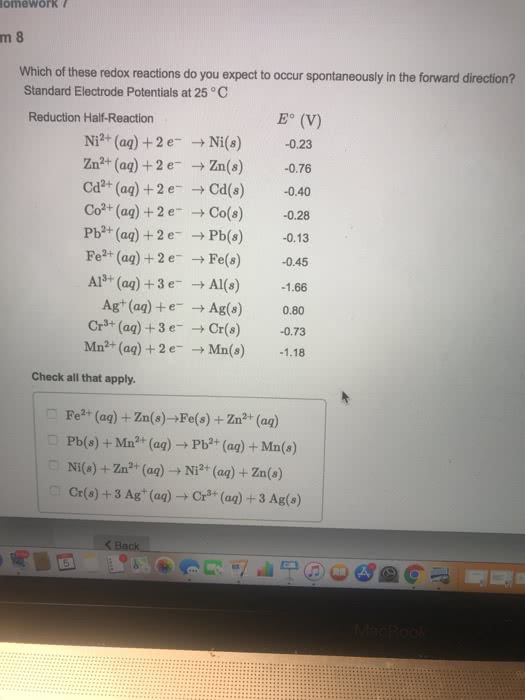
1
answer
0
watching
422
views
20 Mar 2020
Using the standard reduction potentials listed in Appendix E, calculate the equilibrium constant for each of the following reactions at 298 K:
Fe(s) + Ni2+(aq)→ Fe2+(aq) + Ni(s)
Co(s) + 2 H+(aq)→Co2+(aq) + H2(g)
10 Br-(aq) + 2 MnO4-(aq) + 16 H+(aq)→ 2 Mn2+(aq) + 8 H2O(aq) + 5 Br2(l)
Using the standard reduction potentials listed in Appendix E, calculate the equilibrium constant for each of the following reactions at 298 K:
Fe(s) + Ni2+(aq)→ Fe2+(aq) + Ni(s)
Co(s) + 2 H+(aq)→Co2+(aq) + H2(g)
10 Br-(aq) + 2 MnO4-(aq) + 16 H+(aq)→ 2 Mn2+(aq) + 8 H2O(aq) + 5 Br2(l)
Reid WolffLv2
24 May 2020