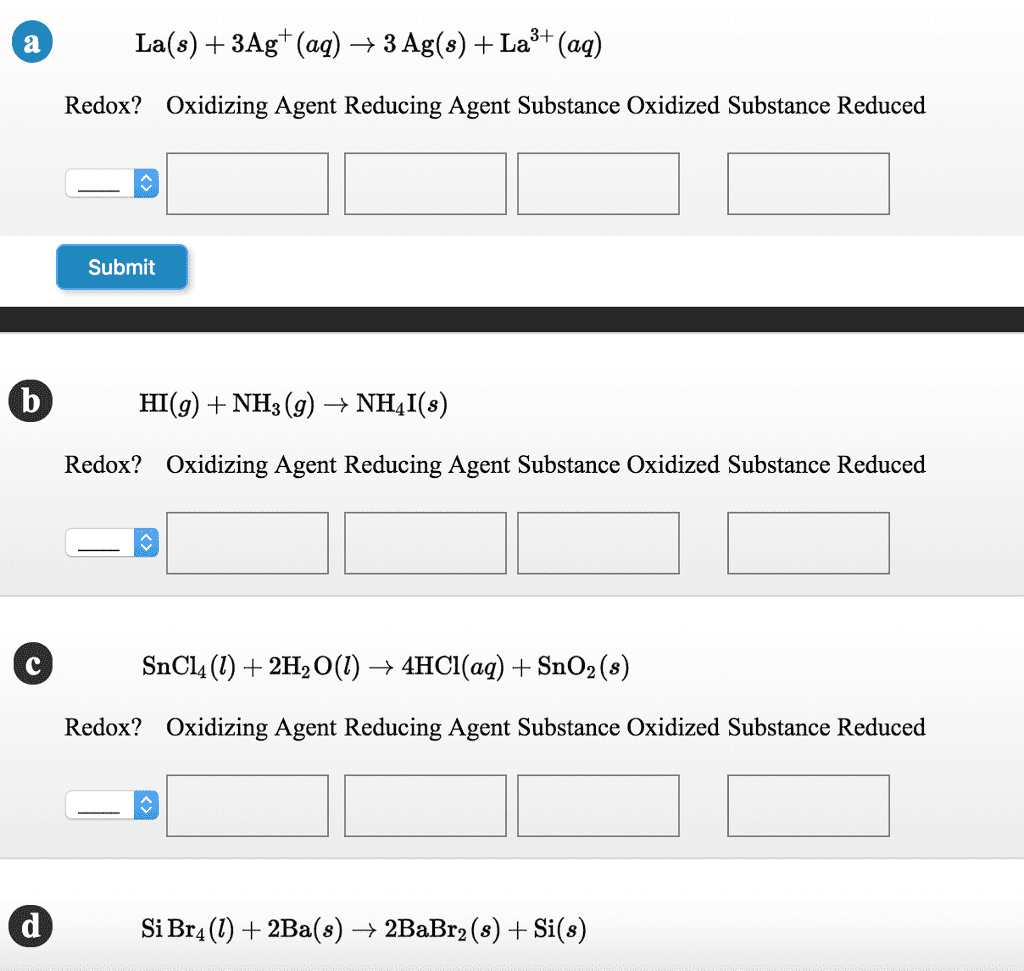
1
answer
0
watching
146
views
19 Mar 2020
Consider the following table of standard electrode potentials for a series of hypothetical reactions in aqueous solution:

Which substance is the strongest oxidizing agent? Which is weakest?
Which substance is the strongest reducing agent? Which is weakest?
Which substance(s) can oxidize C2+?
Consider the following table of standard electrode potentials for a series of hypothetical reactions in aqueous solution:

Which substance is the strongest oxidizing agent? Which is weakest?
Which substance is the strongest reducing agent? Which is weakest?
Which substance(s) can oxidize C2+?
Patrina SchowalterLv2
21 May 2020