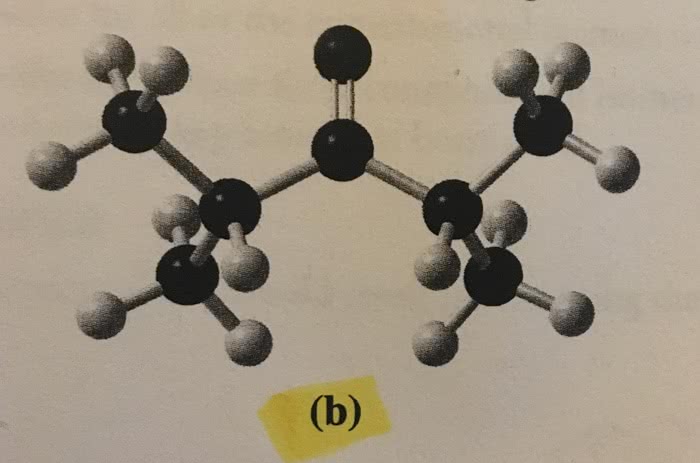1
answer
0
watching
464
views
28 Mar 2020
Butadiene, C4H6, is a planar molecule that has the following carbon–carbon bond lengths:

(a) Predict the bond angles around each of the carbon atoms and sketch the molecule.
(b) Compare the bond lengths to the average bond lengths listed in Table 8.5. Can you explain any differences?
Butadiene, C4H6, is a planar molecule that has the following carbon–carbon bond lengths:
(a) Predict the bond angles around each of the carbon atoms and sketch the molecule.
(b) Compare the bond lengths to the average bond lengths listed in Table 8.5. Can you explain any differences?
Jamar FerryLv2
27 May 2020