1
answer
1
watching
1,215
views
7 Apr 2020
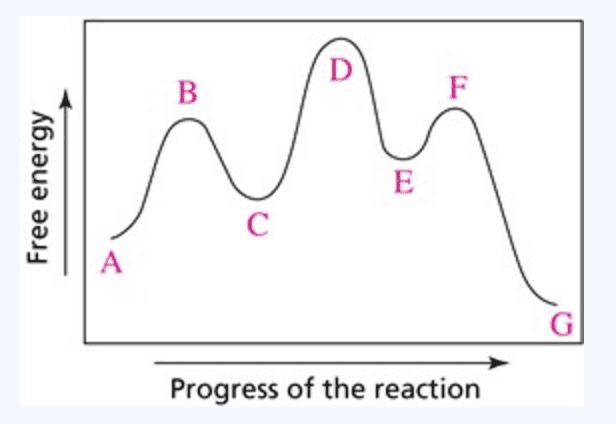
Based on the following reaction profile, how many intermediates are formed in the reaction A  C? How many transition states are there? Which step, A
C? How many transition states are there? Which step, A  B or B
B or B  C, is the faster? For the reaction A
C, is the faster? For the reaction A  C, is ∆E positive, negative, or zero?
C, is ∆E positive, negative, or zero?

Based on the following reaction profile, how many intermediates are formed in the reaction A C? How many transition states are there? Which step, A
B or B
C, is the faster? For the reaction A
C, is ∆E positive, negative, or zero?
Irving HeathcoteLv2
22 May 2020