1
answer
0
watching
638
views
28 Apr 2020
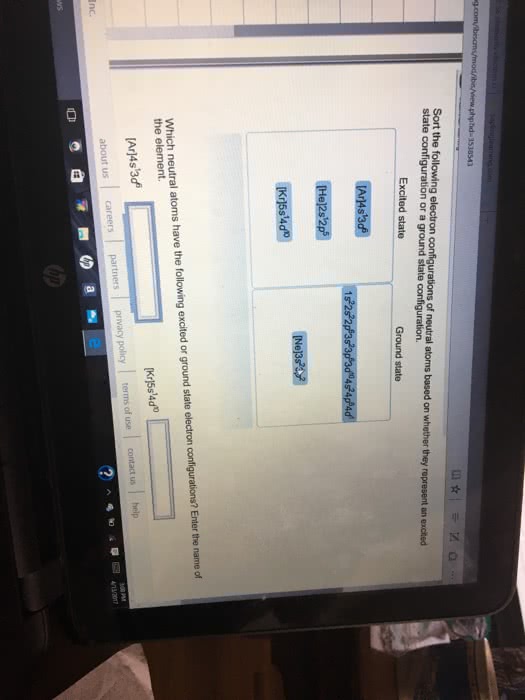
The following electron configurations represent excited states. Identify the element and write its ground-state condensed electron configuration. (a) 1s22s22p43s1, (b) [Ar]4s13d104p25p1, (c) [Kr]5s24d25p1.
The following electron configurations represent excited states. Identify the element and write its ground-state condensed electron configuration. (a) 1s22s22p43s1, (b) [Ar]4s13d104p25p1, (c) [Kr]5s24d25p1.
Liked by standwaiti001
Casey DurganLv2
28 May 2020