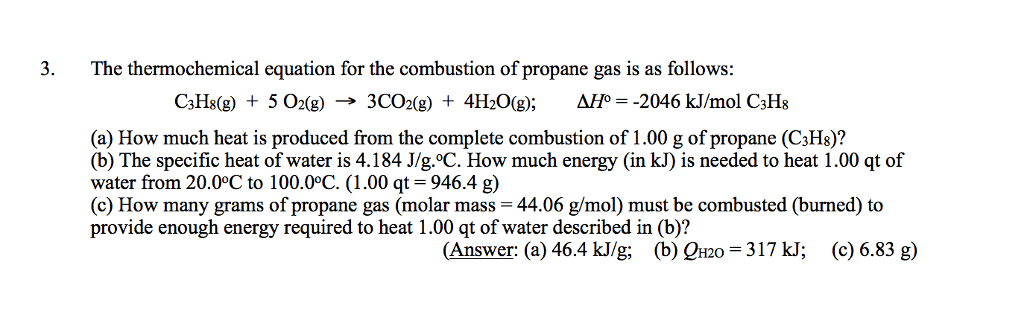1
answer
0
watching
399
views
27 Mar 2020
Many portable gas heaters and grills use propane, C3H8(g), as a fuel. Using standard enthalpies of formation, calculate the quantity of heat produced when 10.0 g of propane is completely combusted in air under standard conditions.
Many portable gas heaters and grills use propane, C3H8(g), as a fuel. Using standard enthalpies of formation, calculate the quantity of heat produced when 10.0 g of propane is completely combusted in air under standard conditions.
Hubert KochLv2
23 May 2020