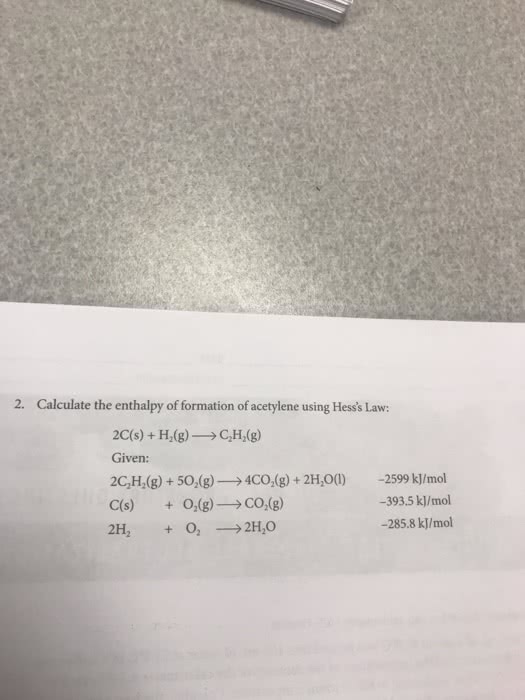1
answer
2
watching
867
views
5 Mar 2020
SECTION/GROUP PRE-LABORATORY QUESTIONS THERMOCHEMISTRY: ACID-BASE NEUTRALIZATION I. Consider this coffee cup calorimetry experiment: a. 100 mL of water at 86.0 C was poured into 100 ml. of water at 23.5°C in a coffee cup Calculate the heat capacity of calorimeter, and the temperature of the contents of the calorimeter equilibrated to 53.5 °C before beginning to fall to room temperature. the calorimeter. (The density and specific heat of water or any aqueous solution can be assumed to be 1.00 g/mL. and 4.184 J/g. "C, respectively)
SECTION/GROUP PRE-LABORATORY QUESTIONS THERMOCHEMISTRY: ACID-BASE NEUTRALIZATION I. Consider this coffee cup calorimetry experiment: a. 100 mL of water at 86.0 C was poured into 100 ml. of water at 23.5°C in a coffee cup Calculate the heat capacity of calorimeter, and the temperature of the contents of the calorimeter equilibrated to 53.5 °C before beginning to fall to room temperature. the calorimeter. (The density and specific heat of water or any aqueous solution can be assumed to be 1.00 g/mL. and 4.184 J/g. "C, respectively)
Nestor RutherfordLv2
13 Mar 2020