1
answer
0
watching
1,011
views
18 Dec 2019
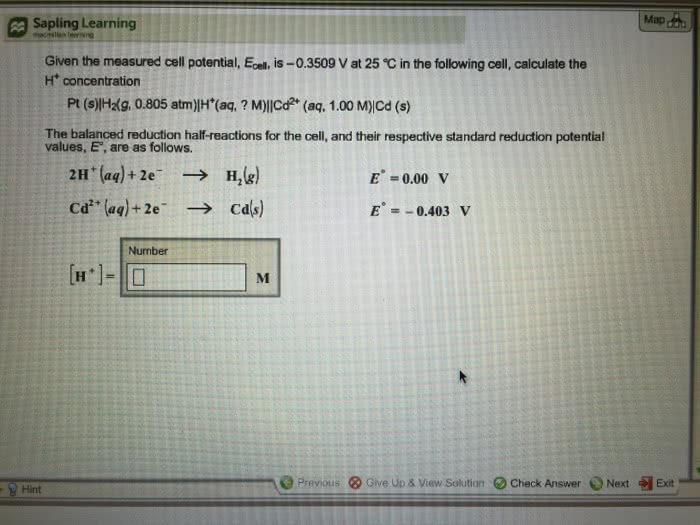
Given the measured cell potential, Ecell, is â0.3653 V at 25 °C in the following cell, calculate the H concentration.
Pt (s)|H2(g, 0.761 atm)|H (aq, ? M)||Cd2 (aq, 1.00 M)|Cd (s)
The balanced reduction half-reactions for the cell, and their respective standard reduction potential values, E°, are as follows.
2H+ (aq)+2e- ---> H2 (g)
Cd2+(aq)+2e- ---> Cd (s)
E0=0.00 v E0=-0.403 v
Given the measured cell potential, Ecell, is â0.3653 V at 25 °C in the following cell, calculate the H concentration.
Pt (s)|H2(g, 0.761 atm)|H (aq, ? M)||Cd2 (aq, 1.00 M)|Cd (s)
The balanced reduction half-reactions for the cell, and their respective standard reduction potential values, E°, are as follows.
2H+ (aq)+2e- ---> H2 (g)
Cd2+(aq)+2e- ---> Cd (s)
E0=0.00 v E0=-0.403 v
Trinidad TremblayLv2
31 Dec 2019