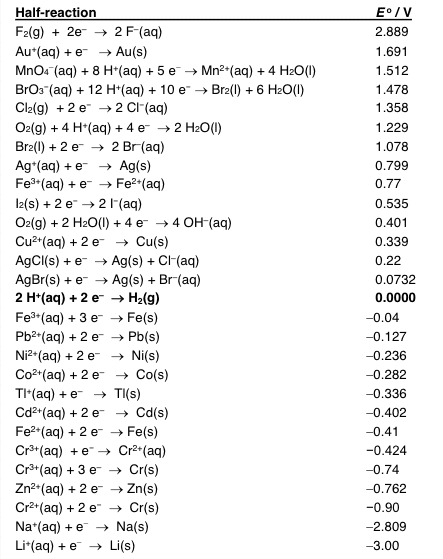
Calculate the expected voltage for each of the following cells (Please show work):
1. Ag, AgCl l Cl- ll Au+3 (all solutions 1.00M)
2. Cd l Cd+2 (0.00050 M) ll Cd+2 (0.69 M) l Cd
3. Fe l Fe+3 (0.056 M) ll Pd+2 (0.64 M) Pd
4. Pt l I- (0.100x1=^-4 M) ll Cr2O7-2 (0.100 M), Cr+3 (0.00100 M), H+ (1.00M) l Pt
Calculate the expected voltage for each of the following cells (Please show work):
1. Ag, AgCl l Cl- ll Au+3 (all solutions 1.00M)
2. Cd l Cd+2 (0.00050 M) ll Cd+2 (0.69 M) l Cd
3. Fe l Fe+3 (0.056 M) ll Pd+2 (0.64 M) Pd
4. Pt l I- (0.100x1=^-4 M) ll Cr2O7-2 (0.100 M), Cr+3 (0.00100 M), H+ (1.00M) l Pt
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
PLS only answer if you know what you're doing and if you are going to answer all of them, i got alot to do cuz of finals so i'd appreciate it if you answer them all correctly, thank you
| 1) Calculate the standard emf (electromotive force) for a voltaic cell whose cell reaction is represented by the balanced equation. 2) Calculate the standard cell potential for a galvanic cell whose cell reaction is represented by the balanced equation. St. Red. Pot. (V)
3) For the voltaic cell shown, calculate the standard cell potential.
|
|