1
answer
1
watching
450
views
13 Dec 2019
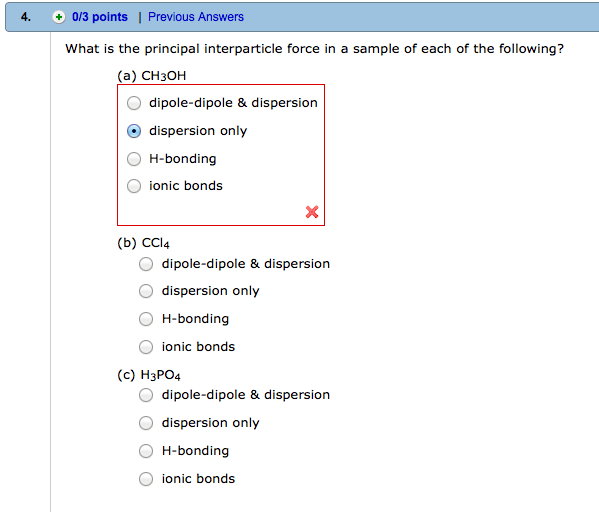
Which of the following statements is false?
The viscosity of a liquid decreases as temperature increases.
The vapor pressure of a substance increases as temperature increases.
The vapor pressure of a substance with strong intermolecular forces is greater than that of a substance with weak intermolecular forces (at the same temperature).
A substance boils when its vapor pressure is equal to the atmospheric pressure.
The boiling point of water increases as atmospheric pressure increases.
2.
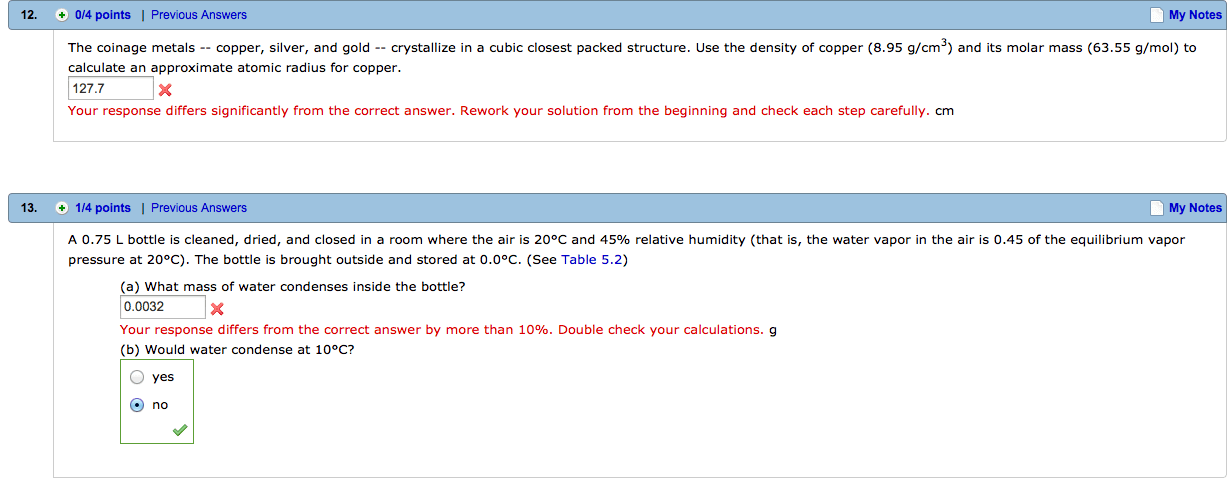
The half-life for a reaction that is first-order in A is 15 minutes. How long will it take for the concentration of A to change from 0.16 M to 0.040 M?
A(g) â B(g) + C(g)
60 min
45 min
30 min
23 min
15 min
Which of the following statements is false?
| The viscosity of a liquid decreases as temperature increases. | |||||||||||||||||
| The vapor pressure of a substance increases as temperature increases. | |||||||||||||||||
| The vapor pressure of a substance with strong intermolecular forces is greater than that of a substance with weak intermolecular forces (at the same temperature). | |||||||||||||||||
| A substance boils when its vapor pressure is equal to the atmospheric pressure. | |||||||||||||||||
| The boiling point of water increases as atmospheric pressure increases. 2. The half-life for a reaction that is first-order in A is 15 minutes. How long will it take for the concentration of A to change from 0.16 M to 0.040 M?
|
Beverley SmithLv2
17 Dec 2019