1
answer
0
watching
238
views
13 Dec 2019
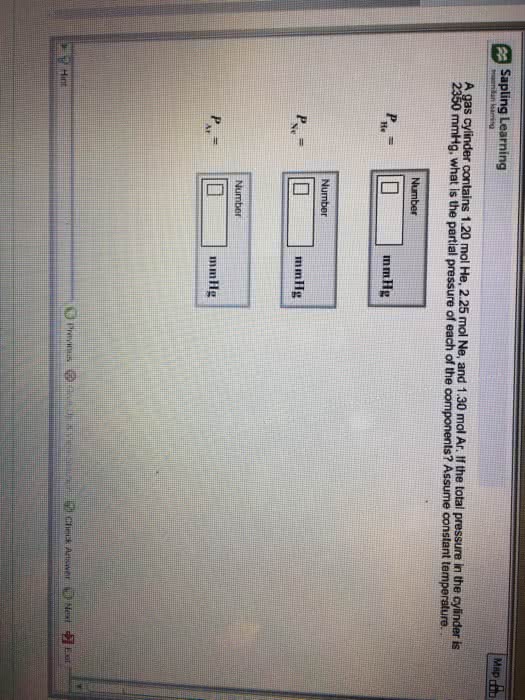
A gas cylinder contains 1.25 mol He, 1.10 mol Ne, and 2.15 molAr. If the total pressure in the cylinder is 2390 mmHg, what is thepartial pressure of each of the components? Assume constanttemperature.
A gas cylinder contains 1.25 mol He, 1.10 mol Ne, and 2.15 molAr. If the total pressure in the cylinder is 2390 mmHg, what is thepartial pressure of each of the components? Assume constanttemperature.
Nelly StrackeLv2
17 Dec 2019