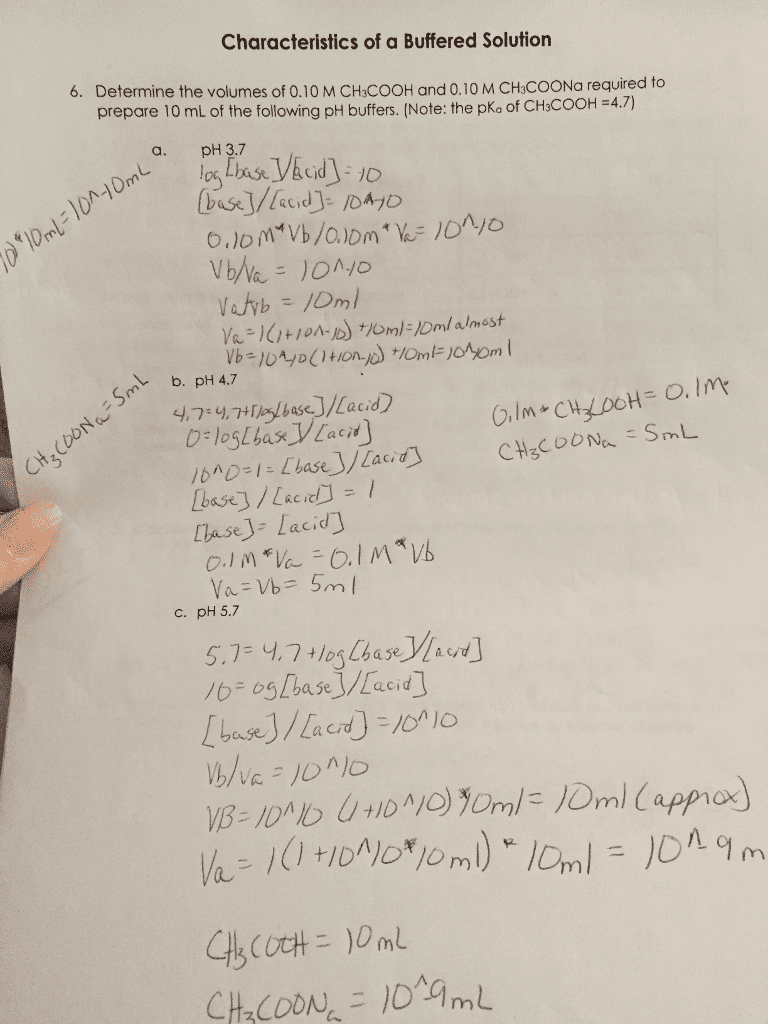0
answers
0
watching
211
views
13 Dec 2019
(a) 0.002 M acetic acid (HAc) (pKa = 4.7) is added to solution. Determine the final pH and [HAc] and [Ac-] in the solution?
(b) How does pH and [HAc] and [Ac-] change when 0.00035 M NaOH, a strong base, is titrated into the solution described in part (a)? Note: NaOH is added from a concentrated stock solution, so the volume change is negligible.
(c) What is the pH and [HCl] and [Cl-] when 2 mM hydrochloric acid (pKa = 0) is added to water?
(d) Discuss whether your results in (a) and (c) are consistent with our assumptions about the dissociation of weak and strong acids.
(a) 0.002 M acetic acid (HAc) (pKa = 4.7) is added to solution. Determine the final pH and [HAc] and [Ac-] in the solution?
(b) How does pH and [HAc] and [Ac-] change when 0.00035 M NaOH, a strong base, is titrated into the solution described in part (a)? Note: NaOH is added from a concentrated stock solution, so the volume change is negligible.
(c) What is the pH and [HCl] and [Cl-] when 2 mM hydrochloric acid (pKa = 0) is added to water?
(d) Discuss whether your results in (a) and (c) are consistent with our assumptions about the dissociation of weak and strong acids.