0
answers
0
watching
186
views
13 Dec 2019
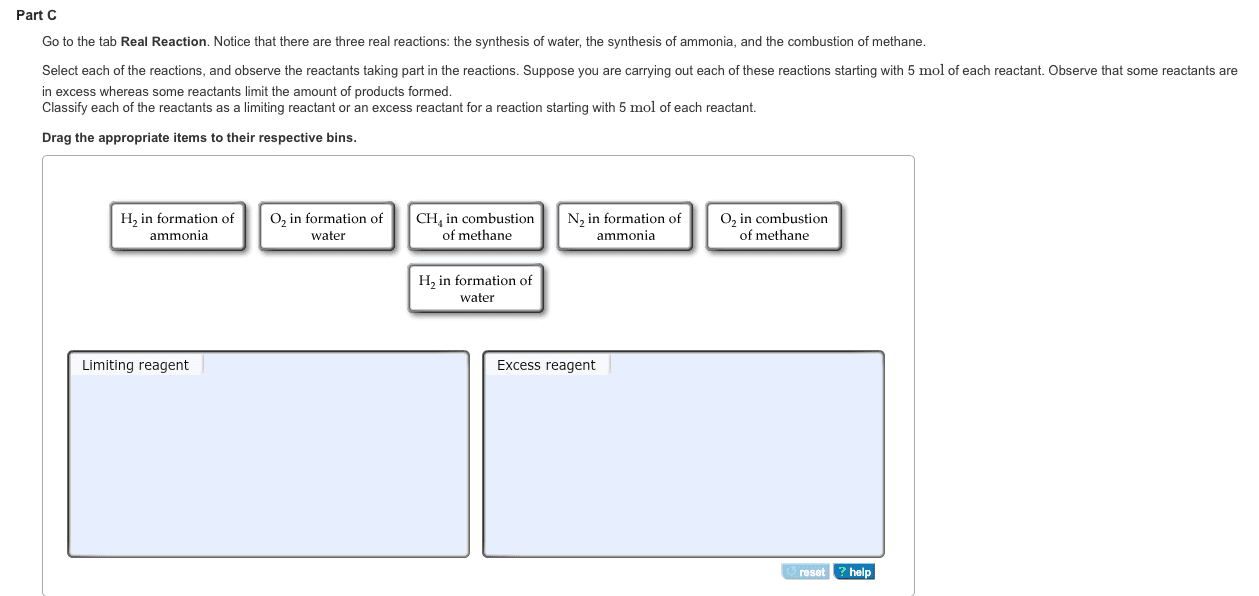
Notice that there are three real reactions: the synthesis of water, the synthesis of ammonia, and the combustion of methane.
Select each of the reactions and observe the reactants taking part in the reactions. Suppose you are carrying out each of these reactions starting with five moles of each reactant. Observe that some reactants are in excess whereas some reactants limit the amount of products formed.
Classify each of the reactants as a limiting reactant or an excess reactant for a reaction starting with five moles of each reactant.
H2 in formation of water O2 in formation of water CH4 in combustion of methane N2 in formation of ammonia H2 in formation of ammonia O2 in combustion of methane
Notice that there are three real reactions: the synthesis of water, the synthesis of ammonia, and the combustion of methane.
Select each of the reactions and observe the reactants taking part in the reactions. Suppose you are carrying out each of these reactions starting with five moles of each reactant. Observe that some reactants are in excess whereas some reactants limit the amount of products formed.
Classify each of the reactants as a limiting reactant or an excess reactant for a reaction starting with five moles of each reactant.
H2 in formation of water
O2 in formation of water
CH4 in combustion of methane
N2 in formation of ammonia
H2 in formation of ammonia
O2 in combustion of methane