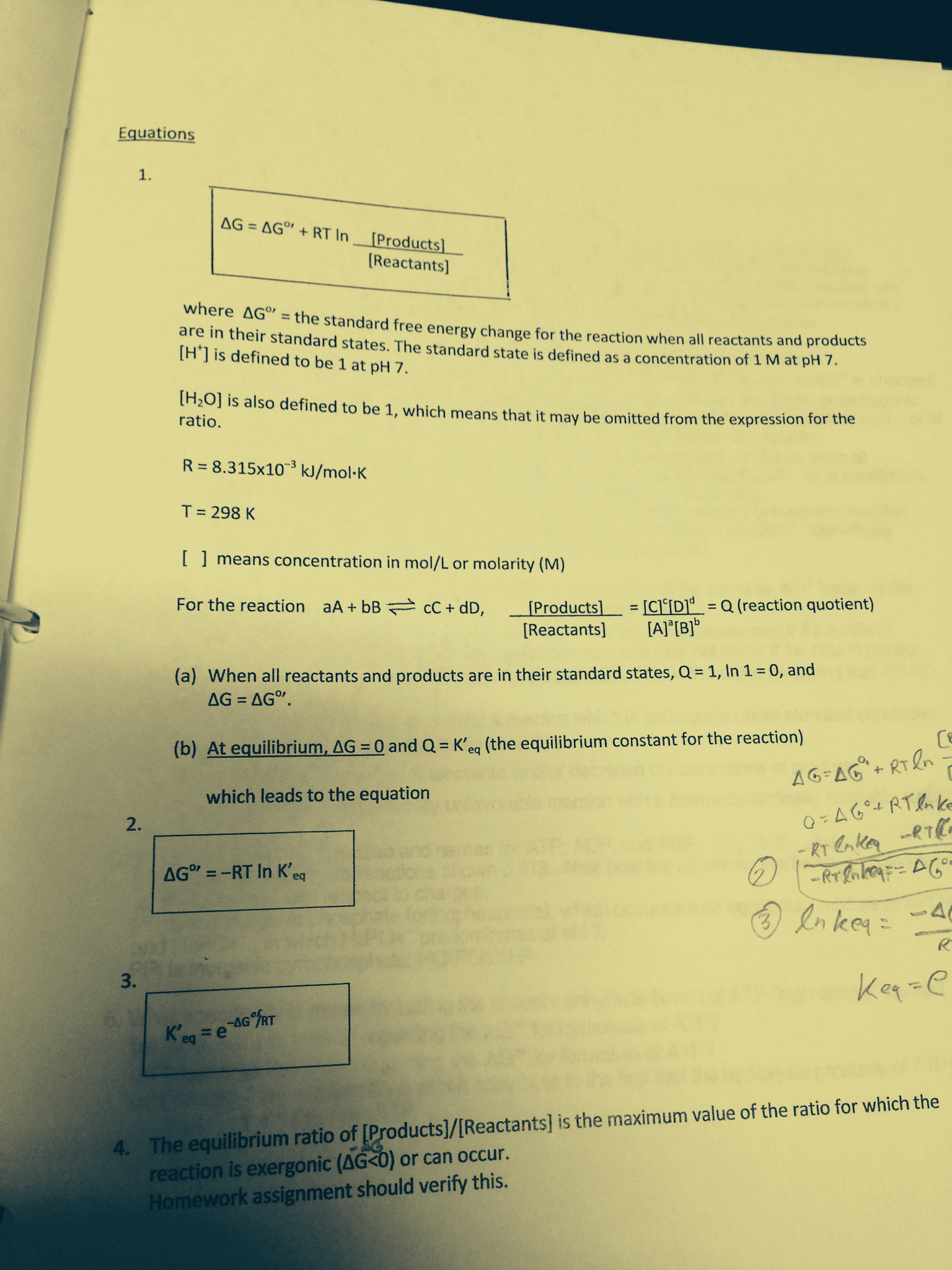In thermodynamics we spend a considerable amount of time determining the energetics of reactions that begin at
âstandard conditionsâ of concentration/pressure. Standard conditions of concentration are exactly 1 M for all aqueous
reactants or products and exactly 1 atm pressure for all gaseous reactants or products.
Answer the following questions assuming we begin (QI) with standard conditions for all reactants and products.
a) Calculate QI under standard conditions of concentration
b) Is the system at equilibrium? (Compare QI to your average Keq)
c) In what direction (if any) will a net reaction take place (fwd. or rev.)?
d) Use a reaction table to find the equilibrium concentrations of reactants and products if we begin at standard
conditions. Note, to solve for final concentrations you will need to use a quadratic equation or the SOLVE function
of your TI calculator. Use your average Keq in the calculation.
In thermodynamics we spend a considerable amount of time determining the energetics of reactions that begin at
âstandard conditionsâ of concentration/pressure. Standard conditions of concentration are exactly 1 M for all aqueous
reactants or products and exactly 1 atm pressure for all gaseous reactants or products.
Answer the following questions assuming we begin (QI) with standard conditions for all reactants and products.
a) Calculate QI under standard conditions of concentration
b) Is the system at equilibrium? (Compare QI to your average Keq)
c) In what direction (if any) will a net reaction take place (fwd. or rev.)?
d) Use a reaction table to find the equilibrium concentrations of reactants and products if we begin at standard
conditions. Note, to solve for final concentrations you will need to use a quadratic equation or the SOLVE function
of your TI calculator. Use your average Keq in the calculation.
For unlimited access to Homework Help, a Homework+ subscription is required.