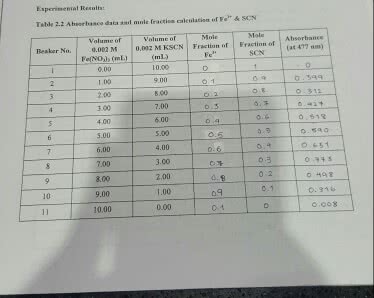
Fe+3 + SCN- <===> FeSCN+2
You mix the following:
Volume of KSCN solution________ 4.35 mL
Original concentration of KSCN solution_________ 0.00321 M
Volume of ferric ion solution ________4.87 mL
Original concentration of ferric ion solution_________ 0.03753M
Voluume of distilled water _______5.25 mL
You measure the %T of the blank and sample at 447 nm using thesame cuvet. Only FeSCN2+ absorbs
at 446 nm! KSCN and Fe+3 are transparent.
%T of blank _____101.7
%T of sample_____ 49.1
constant [Molarity FeSCN2+ per absorbance]______ 0.001756M/A
********NOTE: This constant is only for this pre-lab*******
Calculate:
a) Millimoles of SCN- originally put in solution _______________mmoles
b) Millimoles of Fe+3 originally put in solution _______________mmoles
c) Total volume of solution _______________ mL
d) Molarity of FeSCN2+ at equilibrium _______________ M
e) Millimoles of FeSCN2+ at equilibrium _______________mmoles
f) Molarity of SCN- at equilibrium _______________ M
g) Molarity of Fe+3 at equilibrium _______________ M
h) Calculated equilibrium constant _______________
so far i found that
a)=0.0140 mmol
b)=0.183 mmol
c)=14.47 mL
i know that i have to use and ICE chart but i am not sure on howto calculate the change in moles from the reactants to theproducts?
if i do (0.014 - x) for the KSCN and (0.183 - x) fot theFe^3+and (0+x) for the FeSCN^+2 i have nothing to set it equal to?that what i dont understand
Fe+3 + SCN- <===> FeSCN+2
You mix the following:
Volume of KSCN solution________ 4.35 mL
Original concentration of KSCN solution_________ 0.00321 M
Volume of ferric ion solution ________4.87 mL
Original concentration of ferric ion solution_________ 0.03753M
Voluume of distilled water _______5.25 mL
You measure the %T of the blank and sample at 447 nm using thesame cuvet. Only FeSCN2+ absorbs
at 446 nm! KSCN and Fe+3 are transparent.
%T of blank _____101.7
%T of sample_____ 49.1
constant [Molarity FeSCN2+ per absorbance]______ 0.001756M/A
********NOTE: This constant is only for this pre-lab*******
Calculate:
a) Millimoles of SCN- originally put in solution _______________mmoles
b) Millimoles of Fe+3 originally put in solution _______________mmoles
c) Total volume of solution _______________ mL
d) Molarity of FeSCN2+ at equilibrium _______________ M
e) Millimoles of FeSCN2+ at equilibrium _______________mmoles
f) Molarity of SCN- at equilibrium _______________ M
g) Molarity of Fe+3 at equilibrium _______________ M
h) Calculated equilibrium constant _______________
so far i found that
a)=0.0140 mmol
b)=0.183 mmol
c)=14.47 mL
i know that i have to use and ICE chart but i am not sure on howto calculate the change in moles from the reactants to theproducts?
if i do (0.014 - x) for the KSCN and (0.183 - x) fot theFe^3+and (0+x) for the FeSCN^+2 i have nothing to set it equal to?that what i dont understand