1
answer
0
watching
239
views
13 Dec 2019
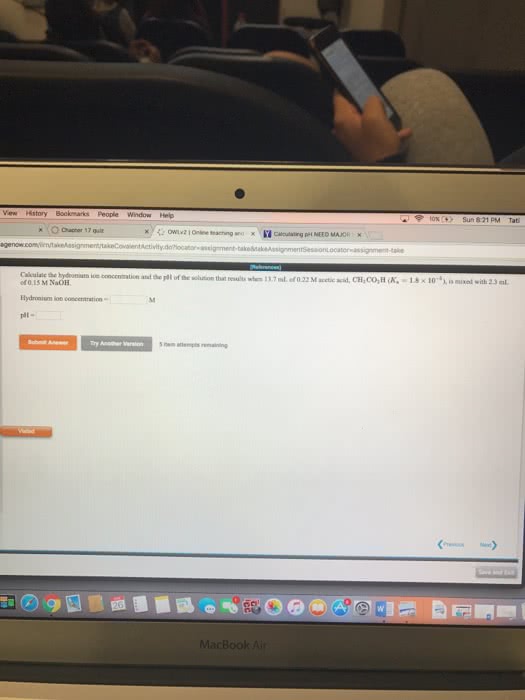
Calculate the hydronium ion concentration and the pH of the solution that results when 24.3 mL of 0.27 M acetic acid, CH3CO2H (Ka=1.8*10^-5), is mixed with 1.1 mL of 0.12 M NaOH.
Hydronium ion concentration = M
pH =
Calculate the hydronium ion concentration and the pH of the solution that results when 24.3 mL of 0.27 M acetic acid, CH3CO2H (Ka=1.8*10^-5), is mixed with 1.1 mL of 0.12 M NaOH.
Hydronium ion concentration = M
pH =
Deanna HettingerLv2
17 Dec 2019