1
answer
0
watching
96
views
13 Dec 2019
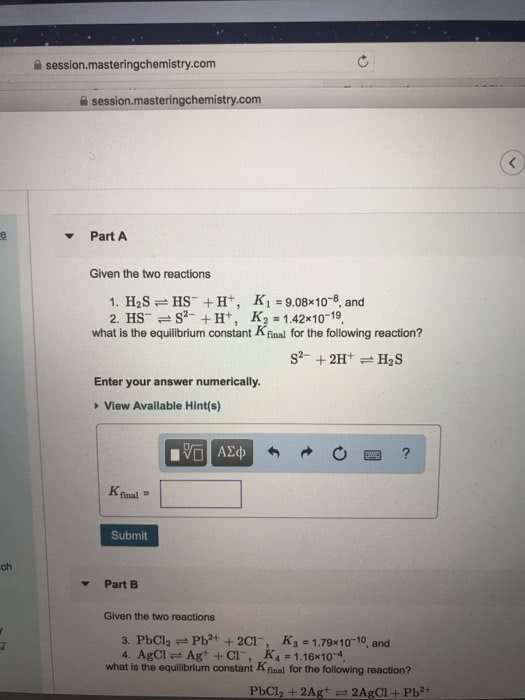
Given the two reactions
PbCl2âPb2++2Clâ, K3 = 1.78Ã10â10, and
AgClâAg++Clâ, K4 = 1.14Ã10â4,
what is the equilibrium constant Kfinal for the following reaction?
PbCl2+2Ag+â2AgCl+Pb2+
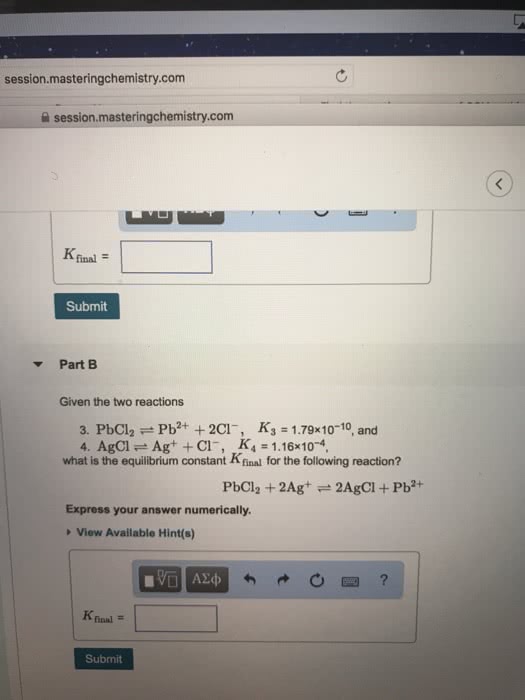
Given the two reactions
PbCl2âPb2++2Clâ, K3 = 1.78Ã10â10, and
AgClâAg++Clâ, K4 = 1.14Ã10â4,
what is the equilibrium constant Kfinal for the following reaction?
PbCl2+2Ag+â2AgCl+Pb2+
Keith LeannonLv2
17 Dec 2019