1
answer
0
watching
147
views
12 Dec 2019
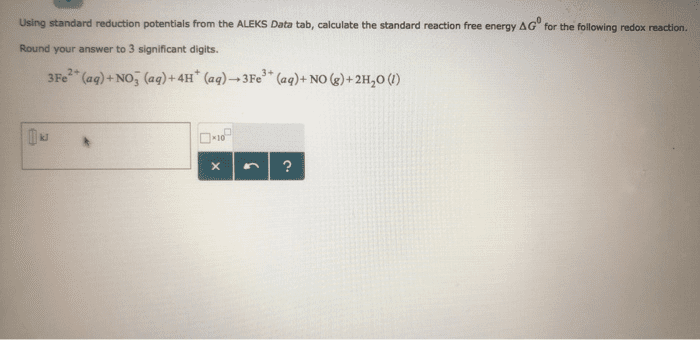
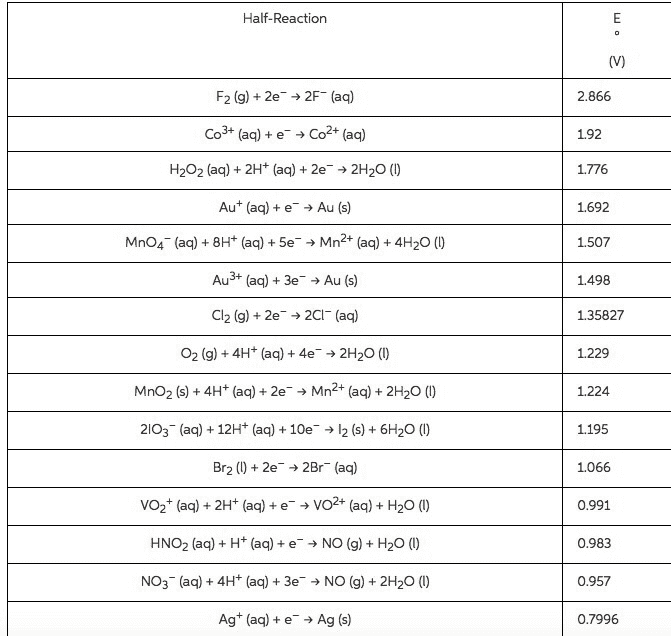
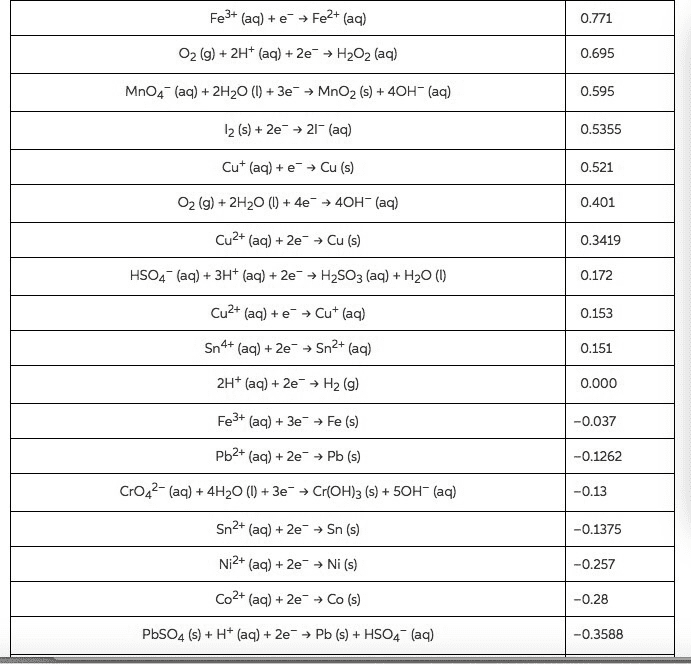
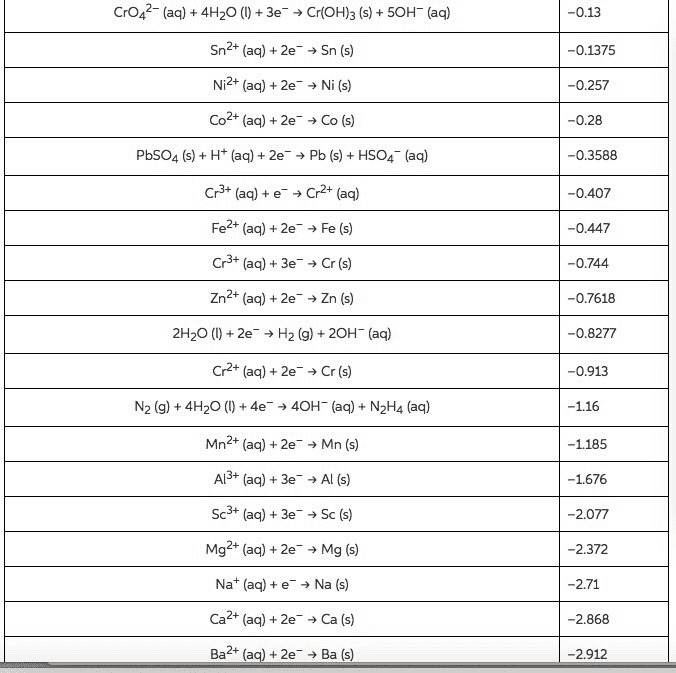
Using standard electrode potentials calculate ÎGârxn and use its value to estimate the equilibrium constant for each of the reactions at 25 âC.
Calculate K
MnO2(s)+4H+(aq)+Cu(s)âMn2+(aq)+2H2O(l)+Cu2+(aq)
Express your answer using one significant figure.
Using standard electrode potentials calculate ÎGârxn and use its value to estimate the equilibrium constant for each of the reactions at 25 âC.
Calculate K
MnO2(s)+4H+(aq)+Cu(s)âMn2+(aq)+2H2O(l)+Cu2+(aq)
Express your answer using one significant figure.
1
answer
0
watching
147
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Tod ThielLv2
13 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
| Exercise 18.65 Use tabulated half-cell potentials to calculate ÎGârxn for each of the following reactions at 25 âC. | Part A Pb2+(aq)+Mg(s)âPb(s)+Mg2+(aq)
SubmitMy AnswersGive Up Part B Br2(l)+2Clâ(aq)â2Brâ(aq)+Cl2(g) Express your answer using two significant figures.
SubmitMy AnswersGive Up Part C MnO2(s)+4H+(aq)+Cu(s)âMn2+(aq)+2H2O(l)+Cu2+(aq) Express your answer using two significant figures.
|