1
answer
0
watching
1,024
views
12 Dec 2019
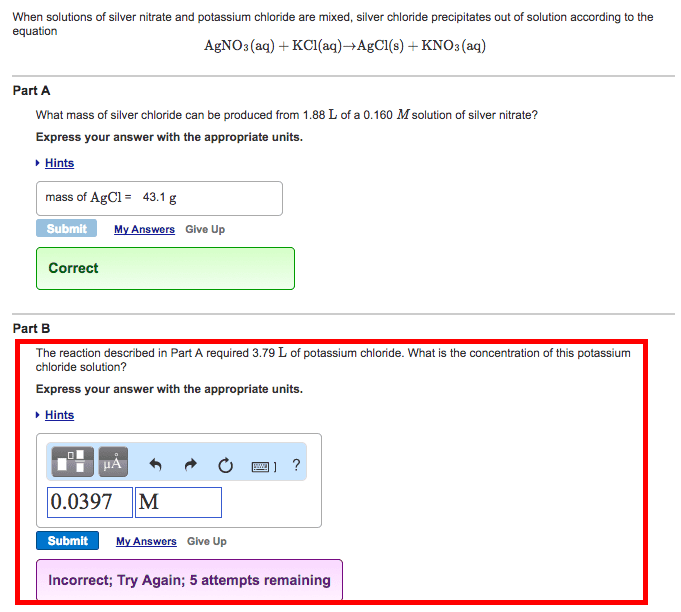
When solutions of silver nitrate and potassium chloride are mixed, silver chloride precipitates out of a solution according to the equation AgNo3(aq)+KCl(aq)-->AgCl(s)+KNO3(aq).
Part A: What mass of silver chloride can be produced from 1.83 L of a 0.213 M solution of silver nitrate? Express your answer with the appropriate units.
The reaction described in Part A required 3.55 L of potassium chloride. What is the concentration of this potassium chloride solution?
Express your answer with the appropriate units.
When solutions of silver nitrate and potassium chloride are mixed, silver chloride precipitates out of a solution according to the equation AgNo3(aq)+KCl(aq)-->AgCl(s)+KNO3(aq).
Part A: What mass of silver chloride can be produced from 1.83 L of a 0.213 M solution of silver nitrate? Express your answer with the appropriate units.
The reaction described in Part A required 3.55 L of potassium chloride. What is the concentration of this potassium chloride solution?
Express your answer with the appropriate units.
Nestor RutherfordLv2
13 Dec 2019