1
answer
0
watching
880
views
12 Dec 2019
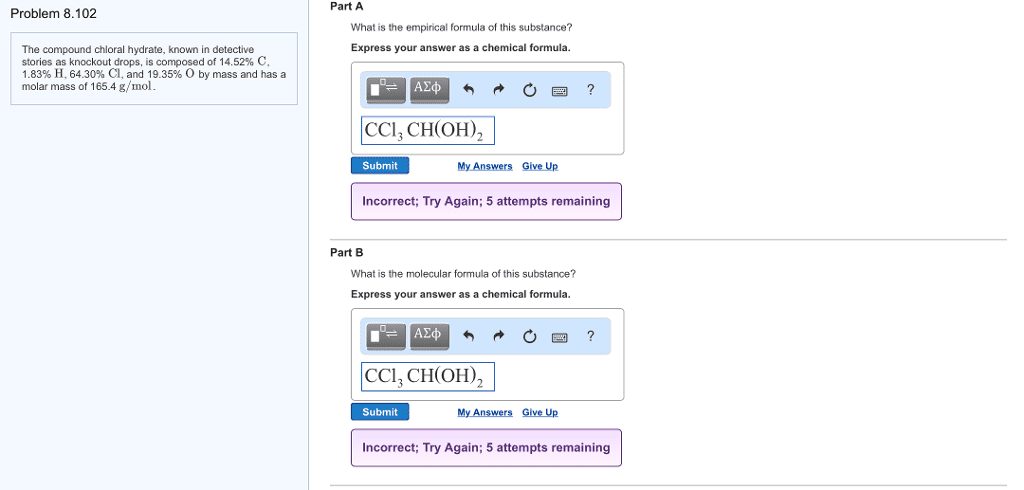
1) A compound with molar mass 110.1 g/mol has the following composition:
C 65.5% H 5.45% O 29.1%
Determine the empirical formula
(Express your answer as a chemical formula.)
(Input the elements in your formula in the order shown above. If parentheses are needed in the formula, be sure to include them in your answer.)
2)
A compound with molar mass 110.1 g/mol has the following composition:
C 65.5% H 5.45% O 29.1%
Determine the empirical formula
(Express your answer as a chemical formula.)
(Input the elements in your formula in the order shown above. If parentheses are needed in the formula, be sure to include them in your answer.)
3)
Arrange the following substances in order of increasing percent of chlorine.
A. KCl B. BaCl2 C. SiCl4 D. LiCl
Enter the letter (A-D) corresponding to each substance.
(least) < < < (greatest)
1) A compound with molar mass 110.1 g/mol has the following composition:
| C | 65.5% |
| H | 5.45% |
| O | 29.1% |
Determine the empirical formula
(Express your answer as a chemical formula.)
(Input the elements in your formula in the order shown above. If parentheses are needed in the formula, be sure to include them in your answer.)
2)
A compound with molar mass 110.1 g/mol has the following composition:
| C | 65.5% |
| H | 5.45% |
| O | 29.1% |
Determine the empirical formula
(Express your answer as a chemical formula.)
(Input the elements in your formula in the order shown above. If parentheses are needed in the formula, be sure to include them in your answer.)
3)
Arrange the following substances in order of increasing percent of chlorine.
| A. | KCl |
| B. | BaCl2 |
| C. | SiCl4 |
| D. | LiCl |
Enter the letter (A-D) corresponding to each substance.
(least) < < < (greatest)
Tod ThielLv2
13 Dec 2019