1
answer
0
watching
102
views
12 Dec 2019
The temperature change during the double replacement reaction between silver (I) nitrate and potassium chloride was measured: AgNO3 (aq) + KCl (aq)-> AgCl(s) + KNO3 (aq) Quantities used: 5.0 mL of 1.35 mol/L of silver (I) nitrate 5.0 mL of 1.45 mol/L potassium chloride. The temperature rise was measured to be 3.1 C. All the heat released was absorbed by the solvent (water). The density of each reacting solution is 1.02 grams/mL. Calculate the total mass of the reacting solutions.
The temperature change during the double replacement reaction between silver (I) nitrate and potassium chloride was measured: AgNO3 (aq) + KCl (aq)-> AgCl(s) + KNO3 (aq) Quantities used: 5.0 mL of 1.35 mol/L of silver (I) nitrate 5.0 mL of 1.45 mol/L potassium chloride. The temperature rise was measured to be 3.1 C. All the heat released was absorbed by the solvent (water). The density of each reacting solution is 1.02 grams/mL. Calculate the total mass of the reacting solutions.
1
answer
0
watching
102
views
For unlimited access to Homework Help, a Homework+ subscription is required.
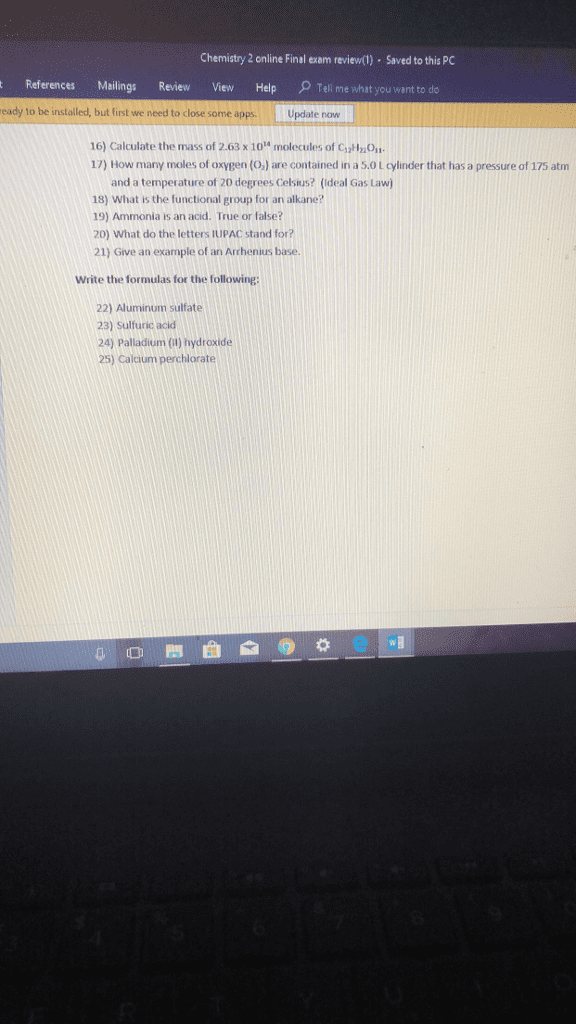
Beverley SmithLv2
13 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232