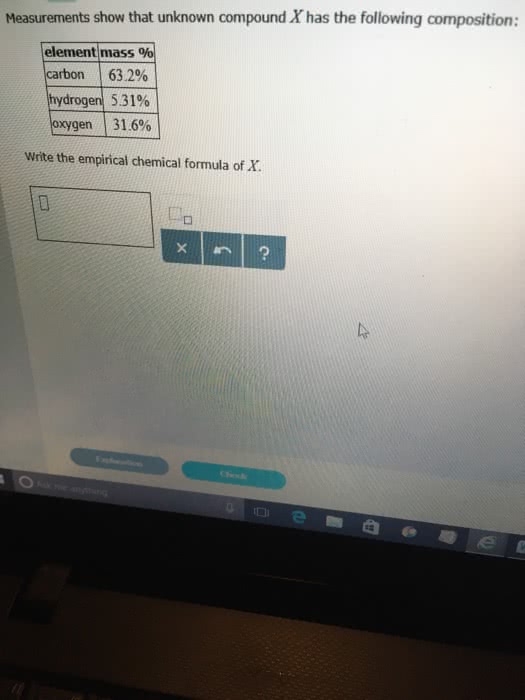
1
answer
0
watching
644
views
11 Dec 2019
An unknown compound has the following chemical formula: Cr(OH)x where x stands for a whole number. Measurements also show that a certain sample of the unknown compound contains 8.0 mol of oxygen and 2.70 mol of chromium. Write the complete chemical formula for the unknown compound.
An unknown compound has the following chemical formula: Cr(OH)x where x stands for a whole number. Measurements also show that a certain sample of the unknown compound contains 8.0 mol of oxygen and 2.70 mol of chromium. Write the complete chemical formula for the unknown compound.
Bunny GreenfelderLv2
13 Dec 2019