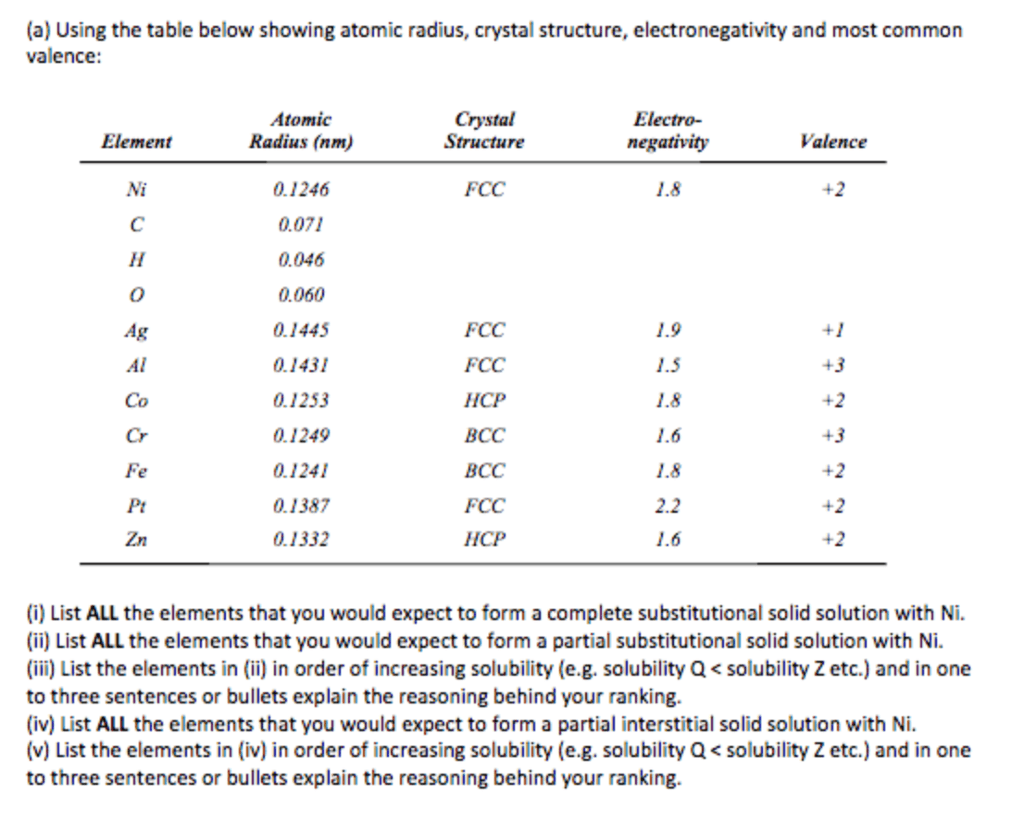
1. Using the data in the table below, answer the questions listed below the table. (5 points total, 1 point each)
Element
Atomic Radius (nm)
Crystal Structure
Electronegativity
Valence
Cu
0.1278
FCC
1.9
+2
Ni
0.1246
FCC
1.8
+2
Cr
0.1249
BCC
1.6
+3
Co
0.1253
HCP
1.8
+2
Fe
0.1241
BCC
1.8
+2
Zn
0.1332
HCP
1.6
+2
Al
0.1431
FCC
1.5
+3
Au
0.144
FCC
2.4
+1
Ag
0.1445
FCC
1.9
+1
Pt
0.139
FCC
2.2
+2
a. Sterling silver is an alloy of 92.5% silver (Ag) and usually copper (Cu). Based on the table above, would you expect complete or partial solid solution for this mix of metals? Why?
b. Zinc is an alloying additive to copper to make brass. Based on the information above, would you expect complete or partial solid solution of zinc in copper? Explain.
c. Would you expect silver and gold to have complete solid solution or limited solid solution? Why?
d. Would you expect iron and chromium to have complete solid solution or limited solid solution? Why?
e. Would you expect chromium and cobalt to have complete solid solution or limited solid solution? Why?
1. Using the data in the table below, answer the questions listed below the table. (5 points total, 1 point each)
| Element | Atomic Radius (nm) | Crystal Structure | Electronegativity | Valence |
| Cu | 0.1278 | FCC | 1.9 | +2 |
| Ni | 0.1246 | FCC | 1.8 | +2 |
| Cr | 0.1249 | BCC | 1.6 | +3 |
| Co | 0.1253 | HCP | 1.8 | +2 |
| Fe | 0.1241 | BCC | 1.8 | +2 |
| Zn | 0.1332 | HCP | 1.6 | +2 |
| Al | 0.1431 | FCC | 1.5 | +3 |
| Au | 0.144 | FCC | 2.4 | +1 |
| Ag | 0.1445 | FCC | 1.9 | +1 |
| Pt | 0.139 | FCC | 2.2 | +2 |
a. Sterling silver is an alloy of 92.5% silver (Ag) and usually copper (Cu). Based on the table above, would you expect complete or partial solid solution for this mix of metals? Why?
b. Zinc is an alloying additive to copper to make brass. Based on the information above, would you expect complete or partial solid solution of zinc in copper? Explain.
c. Would you expect silver and gold to have complete solid solution or limited solid solution? Why?
d. Would you expect iron and chromium to have complete solid solution or limited solid solution? Why?
e. Would you expect chromium and cobalt to have complete solid solution or limited solid solution? Why?