1
answer
0
watching
201
views
11 Dec 2019
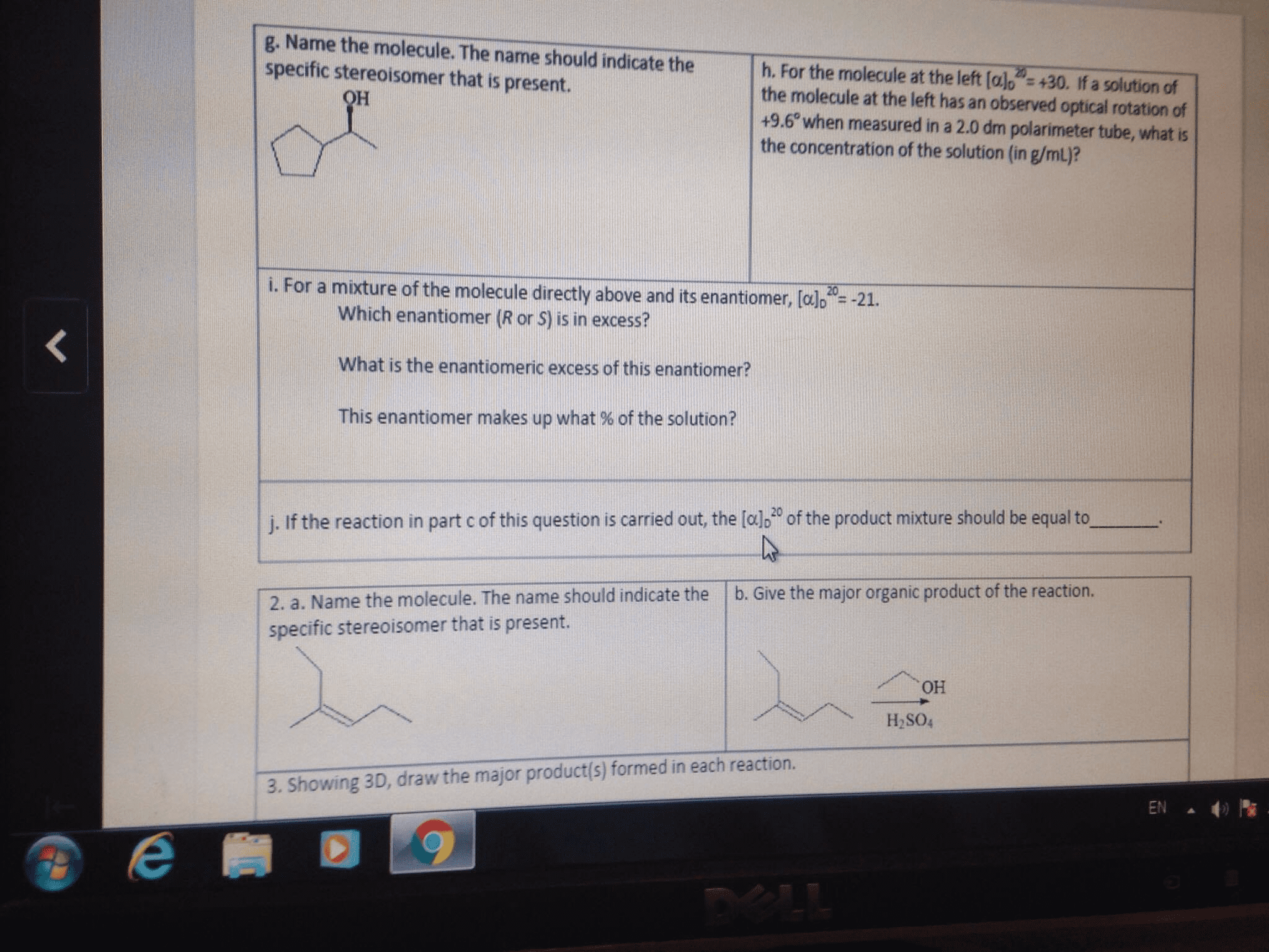
A student is using the polarimeter to analyze a solution containing a mixture of enantiomers. The student observes a rotation of -32.8º. Knowing the cell length is 10.5 cm, the concentration of the solution is 0.79 g/mL, and the specific rotation of the pure (-)-enantiomer is -53.6º, what is the percent enantiomeric excess of the (-)-enantiomer in the student's solution?
| A student is using the polarimeter to analyze a solution containing a mixture of enantiomers. The student observes a rotation of -32.8º. Knowing the cell length is 10.5 cm, the concentration of the solution is 0.79 g/mL, and the specific rotation of the pure (-)-enantiomer is -53.6º, what is the percent enantiomeric excess of the (-)-enantiomer in the student's solution? |
Jean KeelingLv2
13 Dec 2019