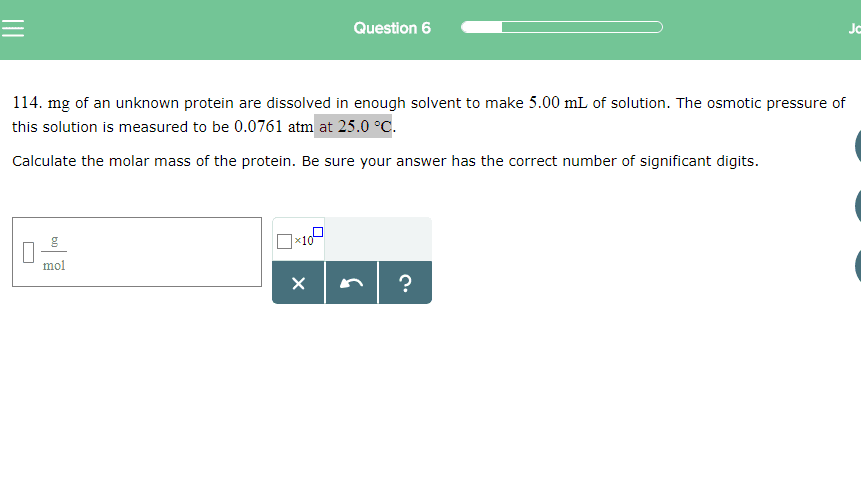
1
answer
0
watching
225
views
11 Dec 2019
Exactly 375 mg of an unknown protein (nonelectrolyte) is dissolved in enough water to give 50.00 mL of solution and the osmotic pressure is measured as 3.25 mmHg at 298 K. Estimate the molar mass of this protein.
Exactly 375 mg of an unknown protein (nonelectrolyte) is dissolved in enough water to give 50.00 mL of solution and the osmotic pressure is measured as 3.25 mmHg at 298 K. Estimate the molar mass of this protein.
Sixta KovacekLv2
13 Dec 2019