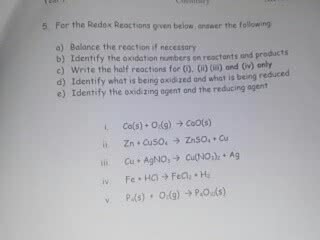1
answer
0
watching
344
views
11 Dec 2019
given the reaction Fe+CuO > FeO+Cu
a. this is what specific type of single displacementreaction?
b. what is oxidized?
c. what is reducated?
d. what is the oxidizing agent?
e. what is the reducing agent?
I have that it is
a. single displacement
b. nothing is oxidized... Fe goes from 0 to -2 -reduced/oxidizing agent
Cu goes from 2+ to 0 - reduced/oxidizing agent
O goes from 2- to 2- - no reaction
c. Fe and Cu are reduced
d. Fe and Cu are the oxidizing agents
e. there is no reducing agents because nothing is oxidized
....PLEASE help i know I went wrong somewhere.
Please show all steps.
given the reaction Fe+CuO > FeO+Cu
a. this is what specific type of single displacementreaction?
b. what is oxidized?
c. what is reducated?
d. what is the oxidizing agent?
e. what is the reducing agent?
I have that it is
a. single displacement
b. nothing is oxidized... Fe goes from 0 to -2 -reduced/oxidizing agent
Cu goes from 2+ to 0 - reduced/oxidizing agent
O goes from 2- to 2- - no reaction
c. Fe and Cu are reduced
d. Fe and Cu are the oxidizing agents
e. there is no reducing agents because nothing is oxidized
....PLEASE help i know I went wrong somewhere.
Please show all steps.
Jean KeelingLv2
13 Dec 2019