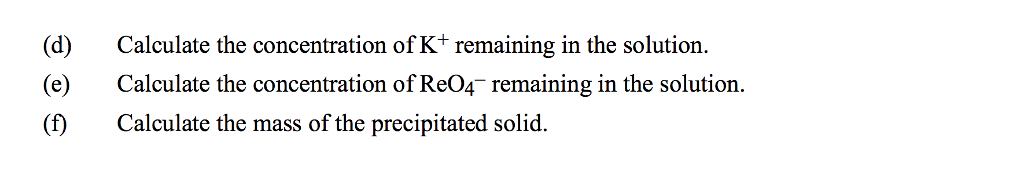1
answer
0
watching
106
views
11 Dec 2019
Can you please explain how you got your anwser?
21. A 1.0 g sample of sugar is added to a beaker containing 100 mL of a saturated solution of sugar. After the solution has come to equilibrium, the resulting sample is filtered to collect all of the undissolved sugar. Assuming constant temperature, the mass of sugar collected is
a) 0.0 g
b) between 0.0 g and 1.0 g
c) 1.0 g
d) more than 1.0 g
Can you please explain how you got your anwser?
21. A 1.0 g sample of sugar is added to a beaker containing 100 mL of a saturated solution of sugar. After the solution has come to equilibrium, the resulting sample is filtered to collect all of the undissolved sugar. Assuming constant temperature, the mass of sugar collected is
a) 0.0 g
b) between 0.0 g and 1.0 g
c) 1.0 g
d) more than 1.0 g
1
answer
0
watching
106
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Nestor RutherfordLv2
13 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232