1
answer
0
watching
120
views
11 Dec 2019
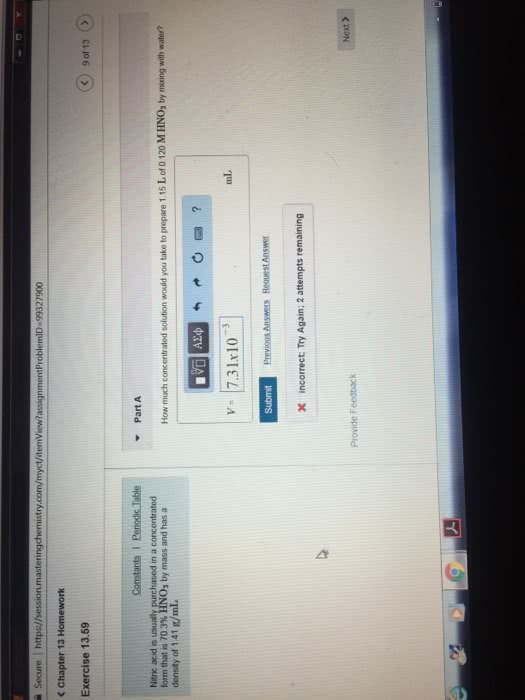
Commercial concentrated nitric acid has a density of 1.4 g / cm 3 and 69% by mass. Considering that M (HNO 3) = 63 g / mol, select from the following options the volume of commercial concentrated nitric acid which, if diluted to 1 L, would generate a solution of nitric acid with
Concentration of 2 mol / L.
A) 130 mL.
B) 125 mL.
C) 65.2 mL.
D) 15.3 mL.
Commercial concentrated nitric acid has a density of 1.4 g / cm 3 and 69% by mass. Considering that M (HNO 3) = 63 g / mol, select from the following options the volume of commercial concentrated nitric acid which, if diluted to 1 L, would generate a solution of nitric acid with
Concentration of 2 mol / L.
A) 130 mL.
B) 125 mL.
C) 65.2 mL.
D) 15.3 mL.
Bunny GreenfelderLv2
13 Dec 2019