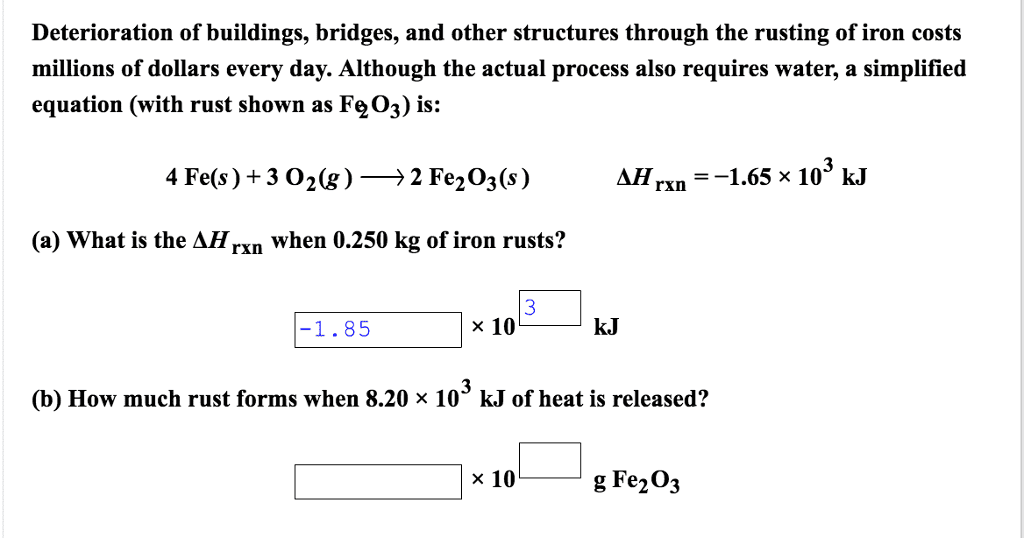1
answer
0
watching
920
views
11 Dec 2019
Be sure to answer all parts.
Deterioration of buildings, bridges, and other structures through the rusting of iron costs millions of dollars every day. Although the actual process also requires water, a simplified equation (with rust shown as Fe2O3) is:
4 Fe(s) + 3 O2(g) â
2 Fe2O3(s) Î
Hrxn = â1.65 Ã
103 kJ
(a) What is the ÎHrxn when 0.250 kg of iron rusts?
Ã
10 kJ
(b) How much rust forms when 6.80 Ã
103 kJ of heat is released?
Ã
10 g Fe2O3
| Be sure to answer all parts. Deterioration of buildings, bridges, and other structures through the rusting of iron costs millions of dollars every day. Although the actual process also requires water, a simplified equation (with rust shown as Fe2O3) is:
(a) What is the ÎHrxn when 0.250 kg of iron rusts? Ã 10 kJ(b) How much rust forms when 6.80 Ã 103 kJ of heat is released?Ã 10 g Fe2O3 |
Elin HesselLv2
13 Dec 2019