Two volatile liquids have vapor pressures of PA* = 525 Torr and PB* = 433 Torr. The liquids are mixed in a solution with composition xA = 0.287. (a) Assuming ideal solution behavior, what would be the partial pressures of A and B in equilibrium with the solution? (b) The actual partial vapor pressures in equilibrium with the solution are PA = 183 Torr and PB = 382 Torr. Calculate the activities and activity coefficients of A and B in this solution.
Two volatile liquids have vapor pressures of PA* = 525 Torr and PB* = 433 Torr. The liquids are mixed in a solution with composition xA = 0.287. (a) Assuming ideal solution behavior, what would be the partial pressures of A and B in equilibrium with the solution? (b) The actual partial vapor pressures in equilibrium with the solution are PA = 183 Torr and PB = 382 Torr. Calculate the activities and activity coefficients of A and B in this solution.
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
Part A
If 0.440 mol of a nonvolatile nonelectrolyte are dissolved in 3.40 mol of water, what is the vapor pressure PH2O of the resulting solution? The vapor pressure of pure water is 23.8 torr at 25 âC .
Express your answer with the appropriate units.
| | |||
| PH2O = |
SubmitHintsMy AnswersGive UpReview Part
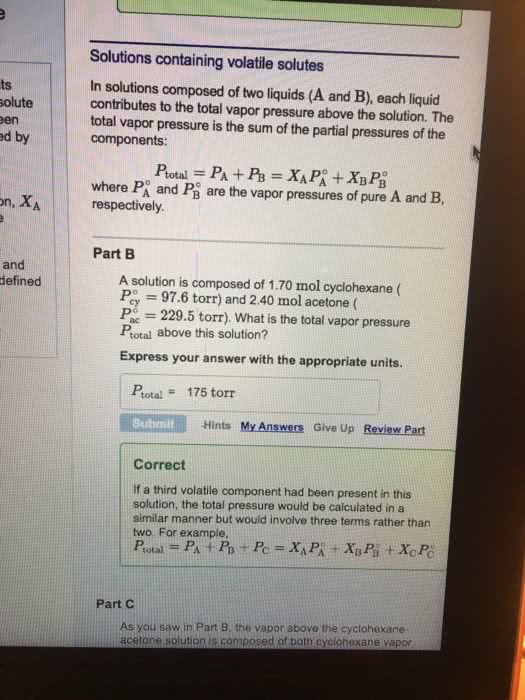
Solutions containing volatile solutes
In solutions composed of two liquids (A and B), each liquid contributes to the total vapor pressure above the solution. The total vapor pressure is the sum of the partial pressures of the components:
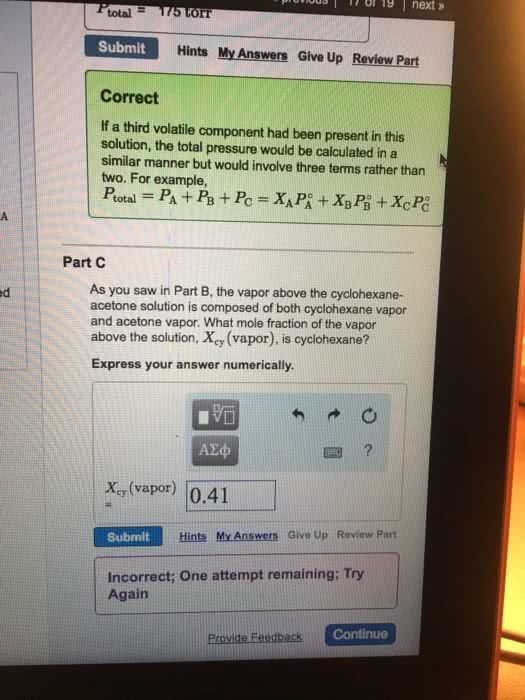
Ptotal=PA+PB=XAPâA+XBPâB
where PâA and PâB are the vapor pressures of pure A and B, respectively.
Part B
A solution is composed of 1.50 mol cyclohexane (Pâcy=97.6 torr) and 2.40 molacetone (Pâac=229.5 torr). What is the total vapor pressure Ptotal above this solution?
Express your answer with the appropriate units.
| | |||
| Ptotal = |