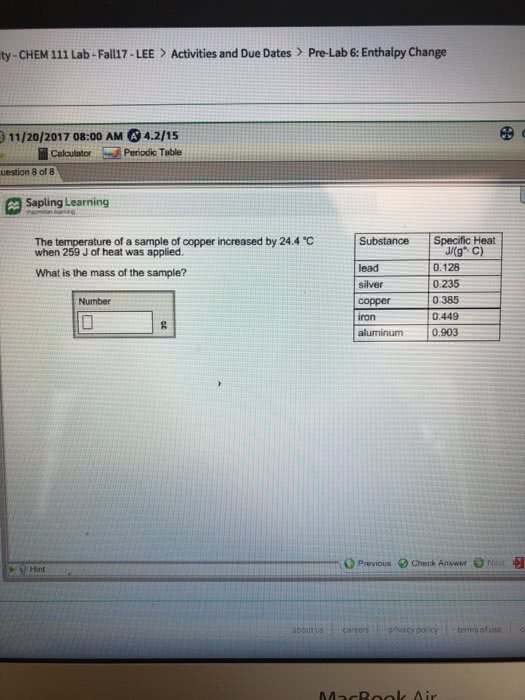
Be sure to answer all questions. Show your work and box your answer, when appropriate. If I cannot read
your handwriting, I cannot give you credit! There is a periodic table on the back of this sheet.
(6) 1. A block of copper weighing 200.0 g absorbs 1.5 kJ of heat. (c = 0.385 J g-1 K-1)
a. What is the temperature change of the copper?
b. If the initial temperature was 25°C, what is the final temperature (in °C)?
(3) 2. A system delivers 495 J of heat and has 825 J of work done on it. Calculate the change in the
internal energy, .Eint, of the system.
(1) 3. Given the following data, which will require more heat to reach the same temperature, a pan made
from aluminum or a pan made from copper (both with the same mass)? (Hint: think about what the
specific heat is actually telling us about the substance.)
Substance
Specific Heat (J g-1 K-1)
Aluminum
0.900
Copper
0.385
Be sure to answer all questions. Show your work and box your answer, when appropriate. If I cannot read
your handwriting, I cannot give you credit! There is a periodic table on the back of this sheet.
(6) 1. A block of copper weighing 200.0 g absorbs 1.5 kJ of heat. (c = 0.385 J g-1 K-1)
a. What is the temperature change of the copper?
b. If the initial temperature was 25°C, what is the final temperature (in °C)?
(3) 2. A system delivers 495 J of heat and has 825 J of work done on it. Calculate the change in the
internal energy, .Eint, of the system.
(1) 3. Given the following data, which will require more heat to reach the same temperature, a pan made
from aluminum or a pan made from copper (both with the same mass)? (Hint: think about what the
specific heat is actually telling us about the substance.)
Substance
Specific Heat (J g-1 K-1)
Aluminum
0.900
Copper
0.385