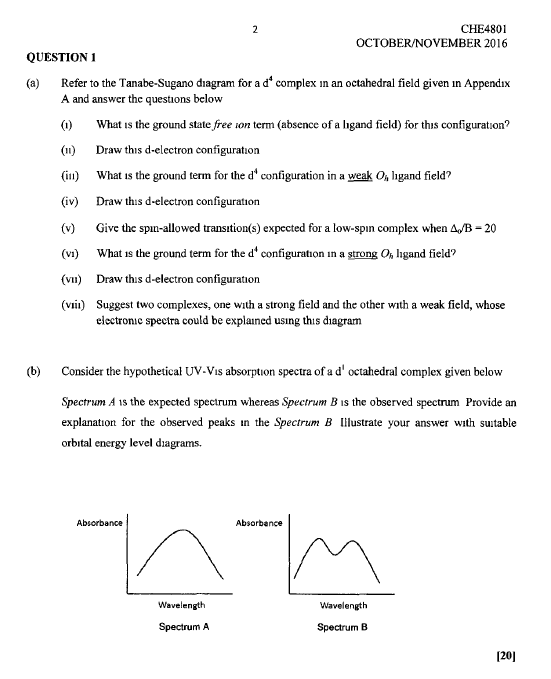
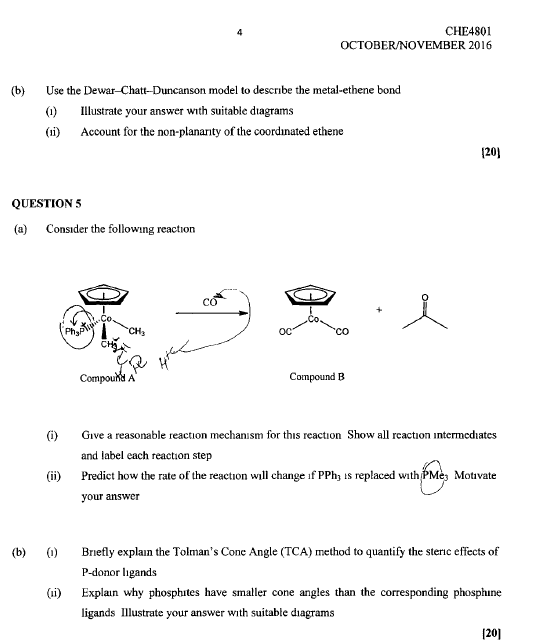
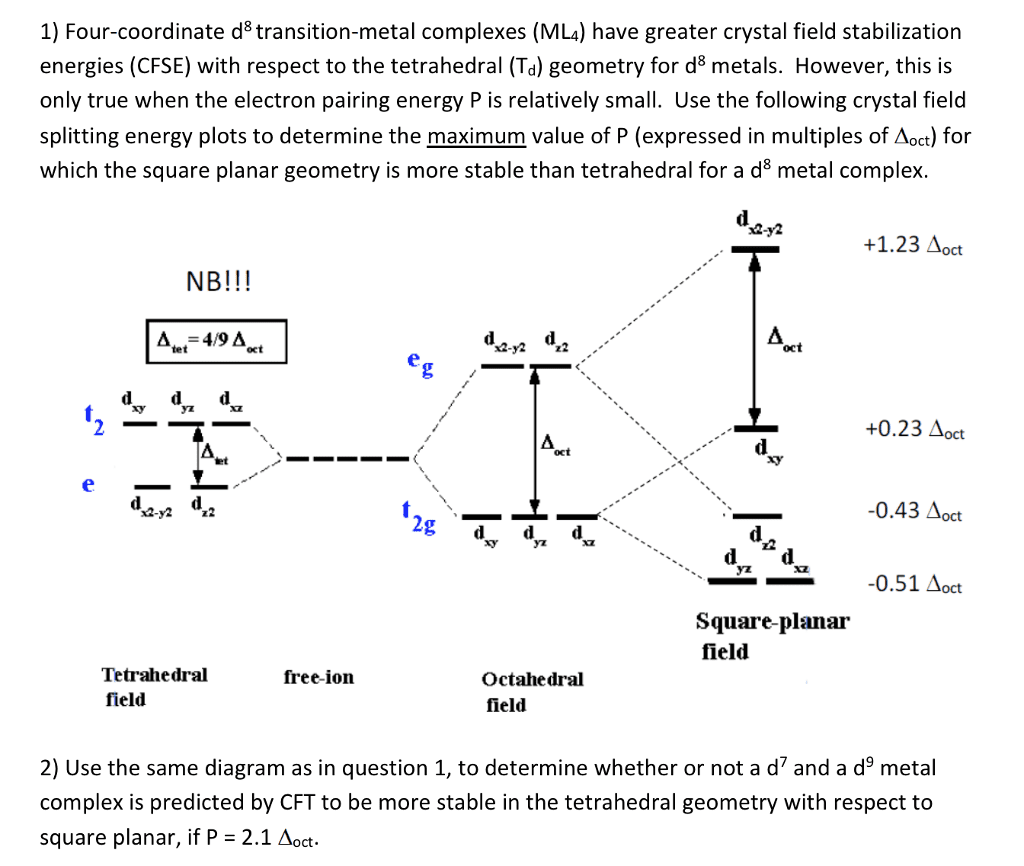
1
answer
0
watching
402
views
11 Dec 2019
Which one of the following statements is INCORRECT?
(A) In an octahedral crystal field, the d electrons on a metal ion occupy the eg set of orbitals before they occupy the t2g set of orbitals.
(B) Diamagnetic metal ions cannot have an odd number of electrons.
(C) Low spin complexes can be paramagnetic.
(D) In high spin octahedral complexes, Îo is less than the electron pairing energy, and is relatively very small.
(E) Low spin complexes are favoured by strong field ligands.
Please explain
Which one of the following statements is INCORRECT?
(A) In an octahedral crystal field, the d electrons on a metal ion occupy the eg set of orbitals before they occupy the t2g set of orbitals.
(B) Diamagnetic metal ions cannot have an odd number of electrons.
(C) Low spin complexes can be paramagnetic.
(D) In high spin octahedral complexes, Îo is less than the electron pairing energy, and is relatively very small.
(E) Low spin complexes are favoured by strong field ligands.
Please explain
Nelly StrackeLv2
13 Dec 2019