1
answer
1
watching
687
views
11 Dec 2019
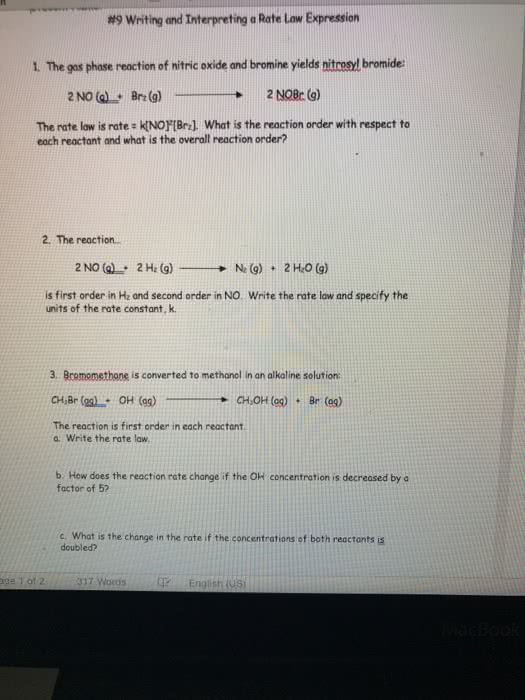
Bromomethane is converted to methanol in an alkaline solution. The reaction is first order in each reactant. CH3Br(aq)+OHâ(aq)âCH3OH(aq)+Brâ(aq)
Write the rate law
How does the reaction rate change if the OHâ concentration is decreased by a factor of 5?
What is the change in rate if the concentrations of both reactants are doubled?
Bromomethane is converted to methanol in an alkaline solution. The reaction is first order in each reactant. CH3Br(aq)+OHâ(aq)âCH3OH(aq)+Brâ(aq)
Write the rate law
How does the reaction rate change if the OHâ concentration is decreased by a factor of 5?
What is the change in rate if the concentrations of both reactants are doubled?
Deanna HettingerLv2
13 Dec 2019