1
answer
1
watching
1,657
views
11 Dec 2019
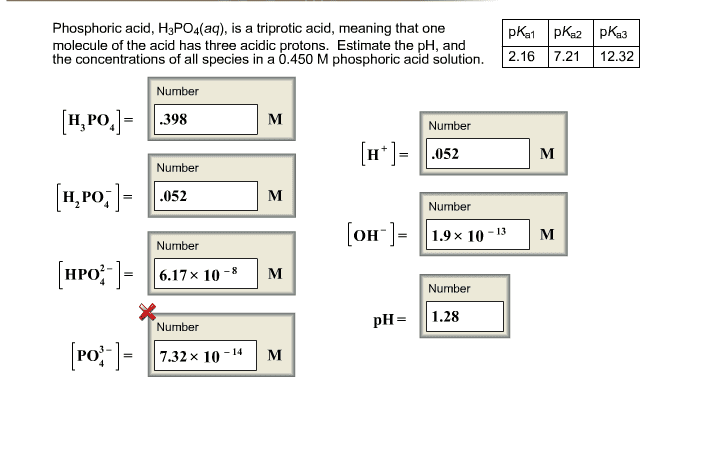
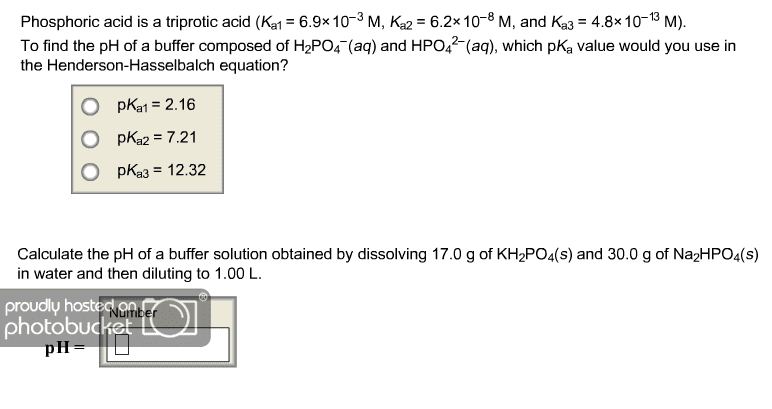
Phosphoric Acid, H3PO4(aq) is a triprotic acid, meaning that one molecule of the acid has three acidic protons. Estimate the pH and the concentrations(M) of all species in a 0.300 M phosphoric acid solution.
pKa1= 2.16 pKa2=7.21 pKa3=12.32
[H3PO4]=
[H2PO4-]=
[HPO4^2-]=
[PO4^3-]
[H+]=
[OH-]=
pH=
Phosphoric Acid, H3PO4(aq) is a triprotic acid, meaning that one molecule of the acid has three acidic protons. Estimate the pH and the concentrations(M) of all species in a 0.300 M phosphoric acid solution.
pKa1= 2.16 pKa2=7.21 pKa3=12.32
[H3PO4]=
[H2PO4-]=
[HPO4^2-]=
[PO4^3-]
[H+]=
[OH-]=
pH=
Casey DurganLv2
13 Dec 2019