1
answer
0
watching
136
views
11 Dec 2019
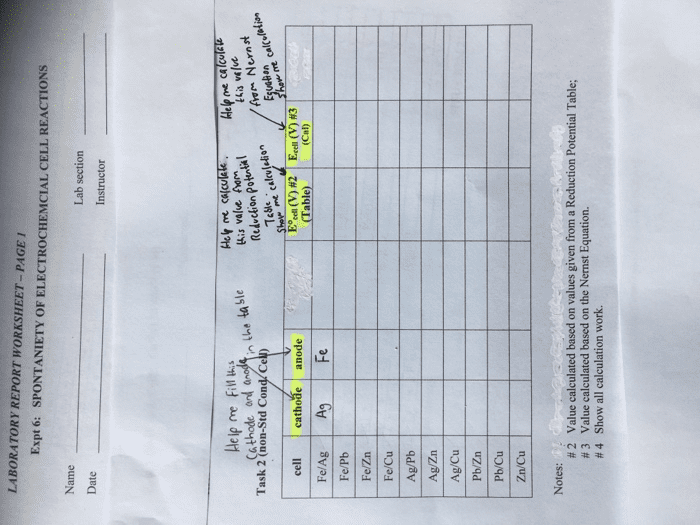
Please fill in the rest of the table below. For theoretical potential, % error, cell reactions for cathode, anode and net, and delta G in kJ.
Table 1
Cell Measured Total
Potential from Multimeter (V)1 Individual Half-Cell Potentials Cell Reactions (anode, cathode, and net) Delta G (kJ) Cu Electrode
Standard Potential (V)2 Metal Electrode
Experimental Potential (V)3 Metal Electrode
Theoretical Potential (V)4 Metal Electrode
Potential % Error Cu Sn 0.419 0.34 -0.079 Cu Al 0.632 0.34 -0.292 Cu Fe 0.461 0.34 -0.121 Cu Zn 0.945 0.34 -0.605
| Please fill in the rest of the table below. For theoretical potential, % error, cell reactions for cathode, anode and net, and delta G in kJ. Table 1 | |||||||
| Cell | Measured Total Potential from Multimeter (V)1 | Individual Half-Cell Potentials | Cell Reactions (anode, cathode, and net) | Delta G (kJ) | |||
| Cu Electrode Standard Potential (V)2 | Metal Electrode Experimental Potential (V)3 | Metal Electrode Theoretical Potential (V)4 | Metal Electrode Potential % Error | ||||
| Cu Sn | 0.419 | 0.34 | -0.079 | ||||
| Cu Al | 0.632 | 0.34 | -0.292 | ||||
| Cu Fe | 0.461 | 0.34 | -0.121 | ||||
| Cu Zn | 0.945 | 0.34 | -0.605 | ||||
1
answer
0
watching
136
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Reid WolffLv2
13 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
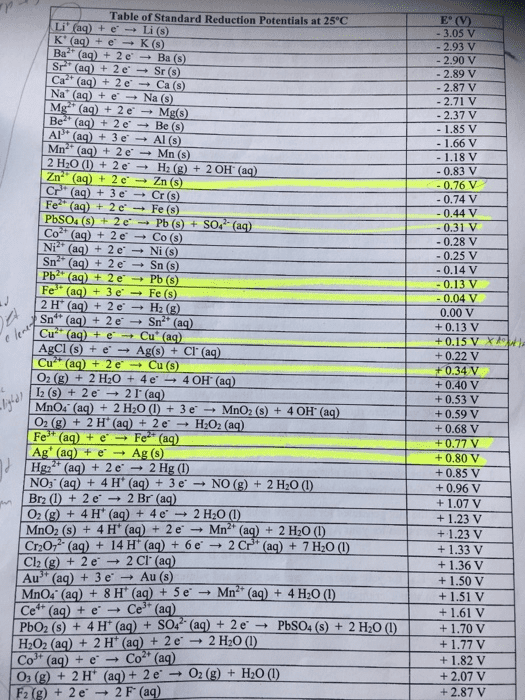
| Free-energy change, ÎGâ, is related to cell potential, Eâ, by the equation ÎGâ=ânFEâ where n is the number of moles of electrons transferred and F=96,500C/(mol eâ) is the Faraday constant. When Eâ is measured in volts and can be determined from half-reaction potentials as given in the table below. ÎGâ must be in joules since 1 J=1 Câ V.
| Part A Calculate the standard free-energy change at 25 âC for the following reaction using the table in the introduction:Mg(s)+Fe2+(aq)âMg2+(aq)+Fe(s) Express your answer numerically in joules.
Part B Calculate the standard cell potential at 25 âC for the reactionX(s)+2Y+(aq)âX2+(aq)+2Y(s) where ÎHâ = -923 kJ and ÎSâ = -123 J/K . Express your answer numerically in volts.
|