3
answers
0
watching
395
views
11 Dec 2019
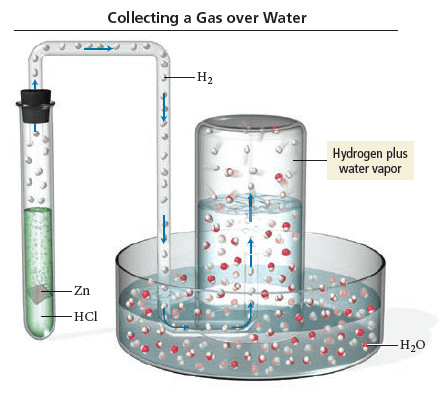
A student carries out a reaction in which 150.1 mL of hydrogen gas is generated and collected over water. The hydrogen gas is produced in a reaction between elemental zinc and an aqueous solution of hydrochloric acid. The total pressure of gases generated and collected in this reaction is 753.7 mmHg at a temperature of 30.0*C. What mass (g) of hydrogen gas was collected in this reaction? (Vapor pressure of water at 30.0*C = 31.86 mmHg)
A student carries out a reaction in which 150.1 mL of hydrogen gas is generated and collected over water. The hydrogen gas is produced in a reaction between elemental zinc and an aqueous solution of hydrochloric acid. The total pressure of gases generated and collected in this reaction is 753.7 mmHg at a temperature of 30.0*C. What mass (g) of hydrogen gas was collected in this reaction? (Vapor pressure of water at 30.0*C = 31.86 mmHg)
10 Jun 2023
Hubert KochLv2
13 Dec 2019
Already have an account? Log in